The loss
of biological diversity is one of the main
consequences of the climate change that humanity
has experienced, becoming a global issue of interest (Arribas et al., 2012). The modification of the landscape, the loss of animal and plant species, the intense
exploitation of natural resources
through intensive agriculture, and the advance of the agricultural frontier result in
the homogenization of the landscape, as
well as the alteration of the microclimate and
of the biogeochemical cycles (i.e., water, carbon,
and nitrogen) (Paruelo et al., 2005). These anthropogenic modifications leave out of
the original habitats, affecting the richness, diversity, and composition of mainly arthropod communities in the soil (Dornelas, 2010). The arthropods, especially the soil macro- arthropods, are an essential component of natural ecosystems and agroecosystems due
to their participation in
regulating fragmentation and
decomposition of organic matter, nutrient recycling (Iannacone and Alvariño, 2006; Coleman et al., 2017). For this reason, the conservation of these organisms is essential due to the multiple services they provide for agroecosystems, which is why they are considered a valuable tool to evaluate the effectiveness of ecosystem management and conservation strategies (Arribas et al., 2012).
Soil arthropods are an essential component of natural and artificial ecosystems due to their adaptability and diversity of species (González-Montaña et al., 2017). They also play an important role in the organic matter decomposition, nutrient recycling, and regulation of biotic communities inside or outside the soil (Wardle, 2006; Brussaard, 2012). Therefore, edaphic arthropods are considered indicators of soil quality and health of any ecosystem (i.e., crops) (Lavelle et al., 2006; Gergócs and Hufnagel, 2009). One of the characteristics that is considered for soil quality is studies of species diversity, mainly explained by the soil spatial heterogeneity that favors the distribution of resources and the coexistence and interaction between species (Wardle, 2006). Also, other studies have revised the relationship between biotic and abiotic components (i.e., physicochemical variables) on soil arthropod diversity (Wardle, 2006; Donoso et al., 2010). For this reason, the generation of information on the direct and indirect effects that agricultural activity can produce on the arthropod communities diversity is vital for the design and improvement of management plans and biological conservation in crops.
Soil physicochemical variables are an essential component for crop development and control; this is because these will depend on soil use (i.e., Permanent crops, Rotation crops, or Pasture). Therefore, the association of soil's physical and chemical properties can be used with the soil composition and abundance of arthropods to develop monitoring and diagnostic tools in local areas (Domínguez et al., 2010). Different studies show that soil factors such as humidity, organic matter content, compaction, agricultural intensification or class of vegetation significantly affect the composition and abundance of edaphic arthropod communities (de Aquino et al., 2008; Diekötter et al., 2010; Feijoo-Martínez et al., 2010; Falco and Momo, 2010).
At present, the municipality of Paipa contributes with 12.1% of the production of peach (Prunus pérsica L) (Espinal et al., 2005), being the experimental farm Tunguavita of the Universidad Pedagógica y Tecnológica de Colombia, one of the leading centers of production of deciduous fruit trees (pears, apples, peaches) in the region. For this reason, this research evaluated the diversity of edaphic arthropods and their association with three soil uses (Permanent crop, Rotation crop, or Pasture) in Tunguavita farm Paipa-Boyacá and to established relationships for the diagnosis of soil quality through biological parameters.
MATERIALS AND METHODS
Area of study. The Tunguavita farm of the Universidad Pedagógica y Tecnológica de Colombia,
located in the municipality of Paipa at an altitude of 2470 m above sea
level, with 5º45' N - 73º06' W, average annual temperature of 14.1 ºC and average relative humidity of 74.9 %. The area has a bimodal rainfall
regime with an average precipitation
of 966 mm, being the sampling near
the beginning of the first rainy season
(Ideam Station No. 2403517). The farm has 134 ha of which three plots corresponding to three different land uses were selected. The first plot corresponds to the permanent cultivation of peach (Prunus pérsica L), Rubidoux variety. The second plot is for rotational use between peach and pear crops between each
season. The third plot corresponds to a pasture area for cattle and sheep. The plots have
less anthropic influence in terms of soil use, making these plots the most stable areas in terms of physicochemical and biological conditions throughout the year.
Field phase. For each land use, sampling was carried out in April 2018. Samples were
taken in a previously selected grid within each plot of 100 x 100 m (1 ha). Within each quadrant, a zigzag path was made, thus choosing a
total of seven sampling points
randomly. At each sampling point,
soil samples were taken for physicochemical and biological analysis
using a 50 x 50 cm grid and a depth
of 10 cm. For the physicochemical
analysis, the soil samples consisted of 3 subsamples in order to obtain a quantity of 200 g of soil. This soil sample was stored in black bags to avoid any
type of degradation due to light and
was sent to the soil laboratory of the Universidad Pedagógica y Tecnológica de Colombia for subsequent physicochemical analysis (IGAC, 2006).
The procedure used for the collection of biological material (edaphic arthropods) was as follows. The sampling points previously selected for the physicochemical variables were taken as a starting point. For each point, with the help of a sieve of 1 mm pore size
and 50 cm in diameter, soil material
was taken to saturation in order to
complete a total of 3 sieving per sampling point. Each individual was collected and separated with entomological forceps and placed in plastic containers to ensure adequate survival conditions during transfer
to the laboratory. According to their visible characteristics, the individuals collected per replicate and location in the laboratory were counted and then grouped into morphospecies. Once the counting was done, one specimen per associated
group was selected to be preserved in 70% alcohol for subsequent taxonomic identification by stereoscopy and taxonomic keys (Vargas et al., 2014; Choate, 2010; Amat- García, 2009).
Laboratory phase. Soil samples were taken from the plots for subsequent analysis. Direct and indirect variables were taken into account in order to have a broader view of the physicochemical variations of the
soil. The analysis of the
physicochemical variables was carried out in the laboratory of the Universidad Pedagógica y Tecnológica de Colombia, with the methods proposed by the Instituto Geográfico Agustín Codazzi (IGAC, 2006) where the following were analyzed: pH
ratio 1:1, %OC Walkey – Black method, %OM =
%OC * 1.724, real density was calculated using the pycnometer method, bulk density was calculated using the
ring of known volume or cylinder, gravimetric humidity percentage (GH) at 105°C for 24 h and volumetric humidity (VH) is the product of the
gravimetric humidity by the value of the bulk density (Forero, 2021)
Statistical analysis. In order to quantify the diversity patterns of edaphic arthropods and the influence of physicochemical variables, the following
statistical analyses were performed for the three sampled plots. The abundance of individuals was determined for each
of the plots, and the abundance and diversity were related to the
three land uses to be visualized by
means of a Ternary graph (Smith, 2017). To determine if significant differences exist within sampling sites for each land use and among the three land uses evaluated, the ANOSIM (Analysis
of Similarities) test was used with a permutation of 999 and a significance level of P< 0.05 (Zubcoff,
2012). Relationships between edaphic
arthropods and soil physicochemical variables were evaluated using a Canonical Correspondence Analysis (CCA) (Ter Braak, 1987). Finally, to evaluate which taxa are mainly responsible for the differences between the three land uses, the SIMPER test was performed with a permutation of 999 and a
significance level of P < 0.05
(Clarke, 1993). All statistical analyses
and tests were performed using the Vegan
library of the Rstudio program (Team R, 2015).
RESULTS AND DISCUSSION
The results showed that organic matter and organic carbon contents were higher in the Permanent crop and Rotation crop,
showing significant differences with the Pasture (P < 0.05). The apparent and real density was higher in the cattle lot but did not differ
significantly from the peach crop
lots (Permanent crop and Rotation crop). Also, gravimetric humidity and volumetric humidity showed significant differences between the Rotation crop
and the other lots. At the same time, the pH levels were higher in the permanent cultivation of
peach (Table 1).
Table 1. Average physicochemical variables for each land use and pairwise test comparing groups; R values and significance levels.
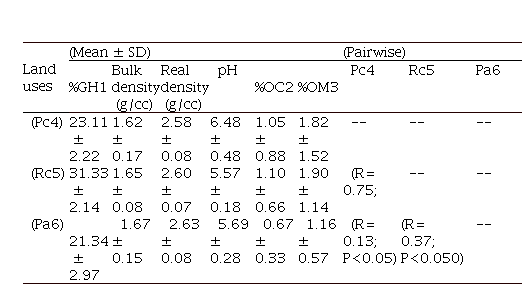 1Gravimetric Humidity; 2Organic carbon; 3Organic matter;
4Permanent crop; 5Rotation crop;
6Pasture.
1Gravimetric Humidity; 2Organic carbon; 3Organic matter;
4Permanent crop; 5Rotation crop;
6Pasture.
|
Land uses
|
(Mean ± SD)
|
(Pairwise)
|
|
%GH1
|
Bulk density (g/cc)
| Real density (g/cc)
| pH |
%OC2 | %OM3 | Pc4 | Rc5 | Pa6 |
|
(Pc4)
|
23.11 ± 2.22
|
1.62 ± 0.17
|
2.58 ± 0.08
|
6.48 ± 0.48
|
1.05 ± 0.88
|
1.82 ± 1.52
|
--
|
--
|
--
|
|
(Rc5)
|
31.33 ± 2.14
|
1.65 ± 0.08
|
2.60 ± 0.07
|
5.57 ± 0.18
|
1.10 ± 0.66
|
1.90 ± 1.14
|
(R= 0.75;
|
--
|
--
|
|
(Pa6)
|
21.34
± 2.97
|
1.67
± 0.15
|
2.63 ± 0.08
|
5.69
± 0.28
|
0.67
± 0.33
|
1.16
± 0.57
|
(R=
0.13; P<0.05)
|
(R= 0.37; P<0.050)
|
--
|
A total of
714 individuals were found in the three
land uses, being (Permanent crop 309, Rotation crop 261, and Pasture 144) distributed
in 8 orders, nine families, and 13 genera (Table 2). The most abundant in Permanent crop and
Rotation crop groups were Haplotaxida, Spirobolida, and Coleoptera, compared to Pasture. In contrast, Aranae, Hymenoptera, and Isopoda were the most abundant only in Pasture (Figure 1). According to the
arthropod group abundance values,
greater diversity in land use was
observed in Permanent crop and Rotation
crop than in Pasture (T-test; P < 0.05). Previous studies have shown that several
arthropod groups are related to soil
organic matter contents, suggesting that resource contribution mainly mediates a better ecosystem status. Thus, the organic matter content
is one of the fundamental axes in the environment
nutrient cycle (Causarano et al., 2007; Coleman and
Wall, 2007; Coleman et al., 2017). Furthermore, soil
physical-chemical characteristics such as pH, texture, compaction, and soil density play an essential role in the preference of the environment for arthropods.
They may even be more relevant than the type of crop or soil use (Galantini and Suñer, 2008). In particular, we find forz all three land uses a very high organic matter input; this can be mainly due to vegetation and fertilization plans, generating an increased availability of energy resources that edaphic invertebrates can rapidly exploit.
Table 2. Taxonomic classification of the edaphic arthropods presents for each of the three land uses.
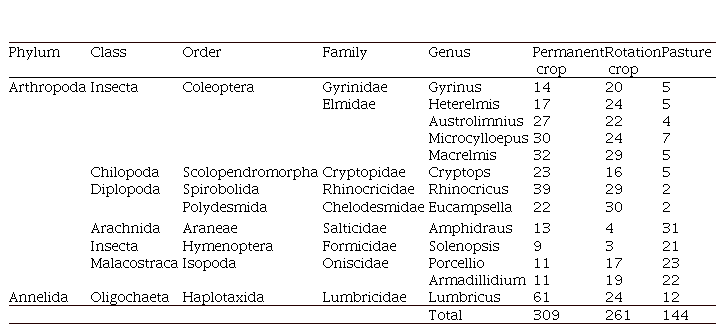
| Phylum | Class | Order | Family | Genus | Permanent crop
|
Rotation
crop
| Pasture |
|
Arthropoda
|
Insecta
|
Coleoptera
|
Gyrinidae
| Gyrinus |
14
|
20
|
5
|
| | |
Elmidae
| Heterelmis |
17
|
24
|
5
|
| | | | Austrolimnius |
27
|
22
|
4
|
| | | | Microcylloepus |
30
|
24
|
7
|
| | | | Macrelmis |
32
|
29
|
5
|
|
Chilopoda
|
Scolopendromorpha
|
Cryptopidae
| Cryptops |
23
|
16
|
5
|
|
Diplopoda
|
Spirobolida
|
Rhinocricidae
| Rhinocricus |
39
|
29
|
2
|
| |
Polydesmida
|
Chelodesmidae
| Eucampsella |
22
|
30
|
2
|
|
Arachnida
|
Araneae
|
Salticidae
| Amphidraus |
13
|
4
|
31
|
|
Insecta
|
Hymenoptera
|
Formicidae
| Solenopsis |
9
|
3
|
21
|
|
Malacostraca
|
Isopoda
|
Oniscidae
| Porcellio |
11
|
17
|
23
|
| | | | Armadillidium |
11
|
19
|
22
|
| Annelida |
Oligochaeta
|
Haplotaxida
|
Lumbricidae
| Lumbricus |
61
|
24
|
12
|
| | | | Total | 309 | 261 | 144 |
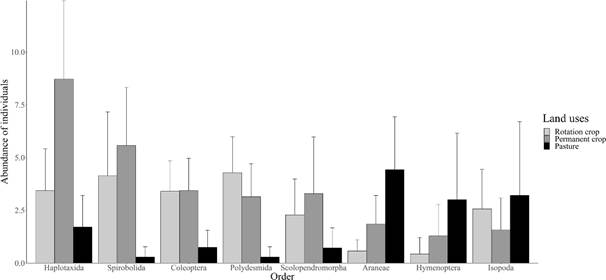 Figure 1. Average abundance of individuals belonging to each Order found at the sampling sites for each of the three land uses.
Figure 1. Average abundance of individuals belonging to each Order found at the sampling sites for each of the three land uses.
The results of Anosim for the biological variables and the physicochemical variables within each of the sampled sub-quadrants were consistent within each of the lands uses used (Anosim: R = 0.19, P = 0.001). Among the land uses it was observed that the biological variables (i.e., arthropod composition) showed significant differences in at least one of the three land uses evaluated (Anosim: R = 0.79, P = 0.001), with the Pastureland use being the most different from the others (Figure 2). This could indicate that the composition and abundance of arthropods would be related not only to intrinsic crop conditions (plant cover), but also to extrinsic factors (i.e., crop management and rotation). Also, the results of Anosim on the physicochemical variables showed significant differences among the three land uses with evidence that the variations found in soil arthropods may correspond to the variations present in each of the land uses evaluated (Anosim: R = 0.58, P = 0.001). In other words, the heterogeneity of the environments in their physicochemical variables and the management plans with different concentrations of chemical or organic fertilizers could impact the nutritional conditions of the soil, which would affect the abundance of soil arthropods present in each land use.
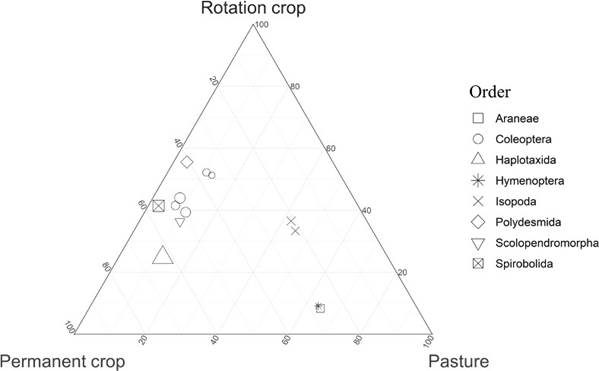 Figure 2. Ternary plot of the groupings of arthropod orders in relation to the three land uses.
Figure 2. Ternary plot of the groupings of arthropod orders in relation to the three land uses.
The total organic matter content showed significant differences between the permanent crop and rotation crop uses with pasture, which is positively related to a greater abundance of edaphic arthropods, this is because they use available organic matter as their primary energy resource (Coleman and Wall, 2007; Coleman
et al., 2017). The type and quality of soil organic matter also condition the availability of nutrients for crops. Factors such as apparent and actual density in Pastureland use resulting from trampling affect the availability of resources and affect mobility under the topsoil (Almada, 2014; Gomez Pamies et al., 2016). While gravimetric, volumetric, and pH
levels may indirectly affect edaphic arthropod communities, these variables depend on the type of vegetation that exists and the management, fertilization, and planning for each land use (Cuesta Segura, 2016; Diaz, 2020).
The
results showed that the type of land use affects the abundance of several groups of edaphic arthropods. For example,
Formicidae was found in greater abundance in the cattle lot, while Chilopoda was only found in the Permanent crop and Rotation crop. The
Oligochaeta group was found in greater abundance in the crop lots of the cattle lot. In contrast, the Diplopoda, Oniscidea, and Araneae
groups have very variable abundance values
among the lots, which can be explained by
the intrinsic factors of each lot (Diekötter et al., 2010; Díaz Porres et al., 2014) or anthropic variables such as insecticides management other chemical compounds. We can explain these results based on two factors: the use of organic matter and soil compaction by cattle as physical-chemical barriers; on the other hand, the influence of present vegetation, lower and less dense, which allows greater ease of movement for walking groups, such as biological barriers. In addition, the application of agricultural inputs such as agrochemicals eliminates other functional groups that live in the soil cover and regulates the edaphic arthropod community (González-Montaña et al., 2017; de Carvalho et al., 2019). This, in turn, contributes to ecological balance because it modifies the
ideal conditions of humidity, aeration,
temperature, pH, and food resources (i.e.,
organic matter) in the top layer of the soil. Therefore, these variations contribute to an environment conducive to the development and adaptation of specialized or highly resilient soil organisms (Bedano et
al., 2016).
The canonical correlation analysis showed an association between physicochemical soil variables and the diversity of taxonomic groups. The factors structure showed that the ratio of %gravimetric humidity, volumetric humidity, and pH
levels are the soil variables that best explain the edaphic arthropods distribution. Correspondence analysis showed a negative association between walking arthropod groups (Araneae and Hymenoptera) with the gravimetric and volumetric soil moisture. While groups of arthropods with elongated bodies
(Coleoptera, Haplotaxida, Scolopendromorpha, Spirobolida, and Polydesmida) were associated with real and apparent soil density values. The
Isopoda (Oniscidae) group was associated with extreme values of soil physicochemical variables, which show that the possible relations between
soil physicochemical and biological variables are a causality of the evaluated plots' intrinsic characteristics.
In addition, the results of the SIMPER test showed the different genera of arthropods contributions in comparison within each of the land uses (Table3). In the comparison of Permanent crop and Rotative crop, it is observed that the genera Lumbricus (Lumbricidae) and Rhinocrius (Rhinocricidae) have a significant contribution (< 0.1%) concerning other genera belonging to groups that may be abundant, such as Cryptops (Cryptopidae). When comparing the land uses of Permanent crop and Pasture, the same pattern is observed in the genera Lumbricus (Lumbricidae) and Rhinocrius (Rhinocricidae) contribution. While comparing Rotative crop and Pasture, we observed that the differences between the two land uses are mainly due to the genus Eucampsella (Chelodesmidae). In general, the results of the SIMPER test showed that the arthropod genera of Permanent crop and Rotation crop contributions are more similar to each other than to the Pasture contributions, showing a possible influence on the joint relationship of physicochemical variables and land use in not only the composition but also in the abundance of large groups of arthropods.
Table 3. SIMPER test results show the contribution and cumulative percentage of Arthropod Genera that have a greater incidence in the differences observed between the three land uses.
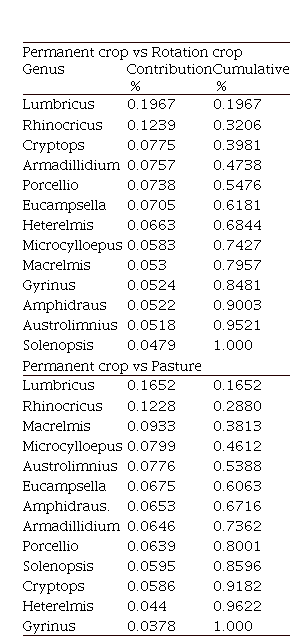
| Permanent crop vs Rotation crop |
| Genus | Contribution% | Cumulative% |
| Lumbricus |
0.1967
|
0.1967
|
|
Rhinocricus
|
0.1239
|
0.3206
|
| Cryptops |
0.0775
|
0.3981
|
| Armadillidium |
0.0757
|
0.4738
|
| Porcellio |
0.0738
|
0.5476
|
| Eucampsella |
0.0705
|
0.6181
|
| Heterelmis |
0.0663
|
0.6844
|
| Microcylloepus |
0.0583
|
0.7427
|
| Macrelmis |
0.053
|
0.7957
|
| Gyrinus |
0.0524
|
0.8481
|
| Amphidraus |
0.0522
|
0.9003
|
| Austrolimnius |
0.0518
|
0.9521
|
| Solenopsis |
0.0479
|
1.000
|
|
Permanent crop vs
Pasture
|
| Lumbricus |
0.1652
|
0.1652
|
| Rhinocricus |
0.1228
|
0.2880
|
| Macrelmis |
0.0933
|
0.3813
|
| Microcylloepus |
0.0799
|
0.4612
|
| Austrolimnius |
0.0776
|
0.5388
|
| Eucampsella |
0.0675
|
0.6063
|
| Amphidraus.
|
0.0653
|
0.6716
|
| Armadillidium |
0.0646
|
0.7362
|
| Porcellio |
0.0639
|
0.8001
|
| Solenopsis |
0.0595
|
0.8596
|
| Cryptops |
0.0586
|
0.9182
|
| Heterelmis |
0.044
|
0.9622
|
| Gyrinus |
0.0378
|
1.000
|
Continued Table 3. SIMPER test results show the contribution and cumulative percentage of Arthropod Genera that have a greater incidence in the differences observed between the three land uses.
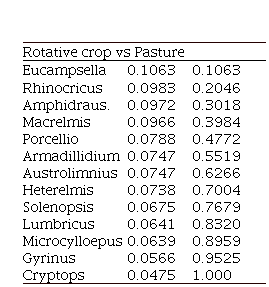
|
Rotative crop vs Pasture
|
| Eucampsella |
0.1063
|
0.1063
|
| Rhinocricus |
0.0983
|
0.2046
|
| Amphidraus.
|
0.0972
|
0.3018
|
| Macrelmis |
0.0966
|
0.3984
|
| Porcellio |
0.0788
|
0.4772
|
| Armadillidium |
0.0747
|
0.5519
|
| Austrolimnius |
0.0747
|
0.6266
|
| Heterelmis |
0.0738
|
0.7004
|
| Solenopsis |
0.0675
|
0.7679
|
| Lumbricus |
0.0641
|
0.8320
|
| Microcylloepus |
0.0639
|
0.8959
|
| Gyrinus |
0.0566
|
0.9525
|
| Cryptops |
0.0475
|
1.000
|
The relationship between the presence and abundance of different soil arthropods with similar designs to the one made here has been studied, and the results are not
univocal (Bedano et al., 2016). Previous work in edaphic mesofauna, mainly in soil mites, showed that the intensity and changes in soil use have an adverse effect on the density of certain
groups (i.e., Oribatide
and Mesostigmata) (Domínguez et al., 2010; Bedano et al., 2016).
Although a typical pattern of
diversity is to observe higher abundances
in land uses with permanent or rotational crops, in some cases, the highest abundances are found in livestock fields in groups belonging to Araneae or
Formicidae as those reported from
elsewhere (Domínguez et al., 2010). The use of techniques such
as "zero tillage" has been
shown to cause a significant decrease in spiders, beetles, and diplopods concerning the lands that are used for
Pasture (Díaz Porres et al., 2014). In our case, the combination of factors, such as changes in
pH, organic matter, compaction, and use of agrochemicals, seems to have a direct effect on the composition and abundance of arthropods and annelids in the soil, despite not being a recurrent effect in some
studies (Moço et al., 2010). These synergistic effects of physicochemical conditions, soil use on species diversity (not abundances), can be mediated mainly by pH, organic matter and organic carbon contents, amount of clays,
sum of bases, and apparent and real density.
Therefore, the environmental heterogeneity hypothesis establishes that the greater the heterogeneity, the greater the number of niches could coexist, resulting in a more
significant number of species but with variable abundances (Allouche et al.,
2012). In this context, and taking into account the soil arthropods diversity presents
in the three land uses, it was observed that
the communities were influenced by the changes
generated in the habitat (land uses), showing that the greatest abundances occur in environments with the most
homogeneous physicochemical conditions. This condition was demonstrated in the permanent crop instead of pasture lot,
which presents fewer environmental modifications.
CONCLUSIONS
The composition and abundance of the taxa allow to quickly evaluate the state of conservation of the soil in the different soil uses, even more, when there is the presence of sensitive organisms (i.e., Oligocheta and Araneae) to the physicochemical or biological barriers.
pH, organic matter and organic carbon contents, amount of clays, the sum of bases, and apparent and real density, seem to be linked not only to the characteristics of each land use but also to the levels of abundance of arthropods that may be found.
The three land uses evaluated in the Tunguavita experimental farm showed different degrees of heterogeneity in the physicochemical variables, which resulted in changes in the soil arthropod community by modifying its diversity in response to the environmental characteristics present; this provides evidence that the soil physicochemical barriers affect the edaphic arthropods composition and diversity present.
Acknowledgments
ACKNOWLEDGEMENTS
The authors thank the Dirección de Investigación of the Universidad Pedagógica
y Tecnológica de
Colombia for its support in carrying out this research. Conflicts of interest: and contributions of authorship. This manuscript was
prepared and reviewed with the participation of the authors, who declare that they do not have any conflict of interest that puts at risk the validity of the
results presented here.
REFERENCES
Allouche, O., Kalyuzhny, M., Moreno-Rueda, G., Pizarro, M., Kadmon, R. 2012. Area–heterogeneity tradeoff and the diversity of ecological communities. Proceedings of the National Academy of Sciences 109(43), 17495-17500. https://doi.org/10.1073/ pnas.1208652109.
Almada, M.S. 2014. Biodiversidad y densidad de arañas (Araneae) en un sistema agropastoril, tendientes a mejorar el impacto de los enemigos naturales sobre insectos plaga. Universidad Nacional de La Plata. https://doi.org/10.35537/10915/34072.
Amat-García, G.D. 2009. Biodiversidad Regional: Santa María, Boyacá. Guía de Campo. Artrópodos: Arácnidos, Miriápodos, Crustáceos, Insectos. Bogotá D C., Colombia. ISBN: 9587193725.
Arribas, P., Abellán, P., Velasco, J., Bilton, D., Lobo, J., Millán, A., Sánchez-Fernández, D. 2012. La vulnerabilidad de las especies frente al cambio climático, un reto urgente para la conservación de la biodiversidad. Ecosistemas, 21(3), 79-84. https://doi.org/10.7818/ECOS.2012.21- 3.10.
Bedano, J.C., Domínguez, A., Arolfo, R., Wall, L.G. 2016. Effect of good agricultural practices under no-till on litter and soil invertebrates in areas with different soil types. Soil and Tillage Research 158, 100- 109. h t t p s : / / d o i . o r g / 1 0 . 1 0 1 6 / j . still.2015.12.005.
Brussaard, L. 2012. Ecosystem services provided by the soil biota. Soil ecology and ecosystem services, 45-58.
Causarano, H.J., Shaw, J.N., Franzluebbers, A.J., Reeves, D.W., Raper, R.L., Balkcom, K.S., Norfleet, M.L. Izaurralde, R.C. 2007. Simulating FieldScale Soil Organic Carbon Dynamics Using EPIC. Soil Sci. Soc. Am. J., 71: 1174-1185.
Choate, P.M. 2010. Introduction to the Identification of Adult Insects and Related Arthropods. University of Florida, Florida, p1-14.
Clarke, K.R. 1993. Nonparametric multivariate analyses of changes in community structure. Australian Journal of Ecology, 18: 117-143.
IGAC, Instituto Geográfico Agustin Codazzi. 2006. Métodos analíticos del laboratorio de suelos. Bogota, p648. ISBN: 9789589067987.
Coleman, D.C., Callaham, M.A., Crossley Jr, D.A. 2017. Fundamentals of soil ecology. Academic press. p376. ISBN: 9780128052518.
Coleman, D.C., Wall, D.H. 2007. Fauna: the engine for microbial activity and transport. Elsevier. 163-191.
Cuesta Segura, A.D. 2016. Respuesta de las comunidades de artrópodos a las perturbaciones ya los cambios en la estructura de la vegetación en ecosistemas de matorral de la Cordillera Cantábrica Arthropod responses to environmental perturbations and changes in the structure of the vegetation in shrub ecosystems of the Cantabrian Mountains. https://doi.org/10.18002/10612/5263.
de Aquino, A.M., da Silva, R.F., Mercante, F.M., Correia, M.E.F., de Fatima Guimaraes, M., Lavelle, P. 2008. Invertebrate soil macrofauna under different ground cover plants in the no-till system in the Cerrado. european journal of soil biology 44(2), 191-197.
de Carvalho, N.L., de Barcellos, A.L., Bubans, V.E. 2019. Ácaros Fitófagos em plantas cultivadas e os fatores que interferem em sua dinâmica populacional. Revista Técnico Científica do IFSC 2(7), 04-17. ISSN: 2316-8382.
Diaz, B.M. 2020. El uso del aliso (Lobularia maritima) para promover artrópodos benéficos en el agroecosistema hortícola. Ediciones INTA. ISBN: 978-987-8333- 33-5.
Díaz Porres, M., Rionda, M.H., Duhour, A.E., Momo, F.R. 2014. Artrópodos del suelo: Relaciones entre la composición faunística y la intensificación agropecuaria. Ecología austral 24(3), 327-334. https://doi.org/10.25260/EA.15.24.3.0.10.
Diekötter, T., Wamser, S., Wolters, V., Birkhofer, K. 2010. Landscape and management effects on structure and function of soil arthropod communities in winter wheat. Agriculture, ecosystems & environment 137(1-2), 108-112.
Domínguez, A., Bedano, J.C., Becker, A.R. 2010. Negative effects of no-till on soil macrofauna and litter decomposition in Argentina as compared with natural grasslands. Soil and Tillage Research 110(1), 51-59. https://doi.org/10.1016/j.still.2010.06.008.
Donoso, D.A., Johnston, M.K., Kaspari, M. 2010. Trees as templates for tropical litter arthropod diversity. Oecologia 164, 201–
Dornelas, M. 2010. Disturbance and change in biodiversity. Philosophical Transactions of the Royal Society of London B: Biological Sciences 365(1558), 3719-3727. http://doi.org/10.1098/rstb.2010.0295.
Espinal, C. F., Martínez Covaleda, H. J., Peña Marín, Y. 2005. La cadena de los frutales de exportación en Colombia: Una mirada global de su estructura y dinámica 1991- 2005. Bogota, p1-65. http://hdl.handle.net/20.500.12324/18862.
Falco, L.B., Momo, F. 2010. Selección de hábitat: efecto de la cobertura y tipo de suelo en lombrices de tierra. Acta zoológica mexicana 26(SPE2), 179-187. ISSN 2448-8445.
Feijoo-Martínez, A., Zúñiga, M.C., Quintero, H., Carvajal-Vanegas, A.F., Ortiz, D.P. 2010. Patrones de asociación entre variables del suelo y usos del terreno en la cuenca del río La Vieja, Colombia. Acta zoológica mexicana, 26(spe2), 151- 164. ISSN 2448-8445.
Forero Pineda, N., Serrano-Cely, P.-A., Forero- Ulloa, F.-E., Almanza-Merchán, P.-J., Cely-Reyes, G.-E. 2021. Composition and abundance of weed-species in relation to physicochemical variables in soil for peach Prunus persica L. var. Rubidoux. Revista Colombiana De Ciencias Hortícolas, 15(2), e12141.
Galantini, J.A., Suñer, L. 2008. Las fracciones orgánicas del suelo: análisis en los suelos de la Argentina. AgriScientia, 25(1), 41–55. htps://doi.org/10.31047/1668.298x.v25. n1.2740.
Gergócs, V., Hufnagel, L. 2009. Application of oribatid mites as indicators. Applied ecology and environmental research 7(1), 79-98. https://doi.org/10.15666/AEER/0701_079098.
Gomez Pamies, D.F., Godoy Guglielmone, M.C., Coronel, J.M. 2016. Macrofauna edáfica en ecosistemas naturales y agroecosistemas de la eco-región Esteros del Iberá (Corrientes, Argentina). págs. 43-56, ISSN 0326-3169.
González-Montaña, L.A., Wilches-Álvarez, W., Rocha-Caicedo, C. 2017. Diversidad alfa y beta de artrópodos epigeos (Arthropoda) en tres sectores sobre la Cordillera Oriental de Colombia. Revista Colombiana de Entomología 43(2), 292-
Iannacone, J., Alvariño, L. 2006. Diversidad de la artropofauna terrestre en la Reserva Nacional de Junín, Perú. Ecología aplicada 5(1-2), 171-174. https://doi.org/10.21704/rea.v5i1-2.332.
Lavelle, P., Decaëns, T., Aubert, M., Barot, S.B., Blouin, M., Bureau, F., Margerie, P., Mora, P., Rossi, J,P. 2006. Soil invertebrates and ecosystem services. European journal of soil biology 42, S3-S15. h t t p s : / / d o i . o r g / 1 0 . 1 0 1 6 / j . ejsobi.2006.10.002.
Moço, M.K.S., Gama-Rodrigues, E.F., Gama- Rodrigues, A.C., Machado, R.C.R., Baligar, V.C. 2010. Relationships between invertebrate communities, litter quality and soil attributes under different cacao agroforestry systems in the south of Bahia, Brazil. Applied soil ecology 46(3), 347-354. https://doi.org/10.1016/j. apsoil.2010.10.006.
Paruelo, J.M., Guerschman, J.P., Verón, S.R. 2005. Expansión agrícola y cambios en el uso del suelo. Ciencia hoy 15(87), 14-23.
Smith, Martin R. 2017. Ternary: An R Package for Creating Ternary Plots. Zenodo, https://10.5281/zenodo.1068996.
Team R. 2015. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. Available online at https://www.R-project.org/.
Ter Braak, C.J.F. 1987. The analysis of vegetation-environment relationships by canonical correspondence analysis. Vegetatio 69, 69–77 https://doi.org/10.1007/BF00038688.
Vargas, J.G.P., Recamier, B.E.M., De Oyarzabal, A. 2014. Guía ilustrada para los artrópodos edáficos. La prensa de Ciencias, Mexico. p84. ISBN: 9786070250712.
Wardle DA. 2006. The influence of biotic interactions on soil biodiversity. Ecol Lett. 9(7):870-86. https://doi.org/10.1111/j.1461- 0248.2006.00931.x. PMID: 16796577.
Additional information
Conflict of Interest: The manuscript was prepared and reviewed with the participation of the authors, who declare that there exists
no conflict of interest that puts at
risk the validity of the presented results.











