INTRODUCTION
Arrow cane (Gynerium
sagitatum Aubl., 2x = 2n = 72) is a Poaceae species whose plants in the
Americas grow between 0 and 1700 (masl) from Central
America, the Antilles to Bolivia and Paraguay. In Colombia, arrow cane plants
grow in the West Planes, Central Andean region and the Caribbean Coast where it
is cared by aboriginal communities established in the flatlands of Córdoba and
Sucre departments since ancient times (GRIN, 2019; Suárez,
2019a). Arrow cane plants are perennial, giant rhizomatous, reed grass
with low part of the culms clothed with bladeless sheaths and upper part with
unfold leaf blades and open fan-shaped form. Culms range from 5-14 m long and
die after flowering. Leaves are bright green 160-230 cm long and 8-14 cm wide;
culms can form as much as 200 leaves during life time with 19-28 fresh blades at
any time (Kalliola, et al.,
1992). Aerial stems are underground
interconnected by a net of leafy rhizomes with shoots at the end point that
grow at a distance 15-20 cm from the original culm. Both, aerial and
underground, structures serve as colonizers of new territories at the same time
that prevent soil erosion (Contreras, et
al., 1998; Kalliola, et al., 1992; Suárez, et al., 2013). The leaf central nerve is the raw material used
by natives to make a colorful variety of worldwide known handicraft, included “Sombrero Vueltiao”
raised as Colombian Cultural Symbol by Congress: Arrow cane crafts are considered
an antique mechanism for religious, artistic and political expression and a
legacy of the Zenú tribe of Indians (Artesanías de
Colombia, 2019).
Large scale cultivation of arrow cane plants to
produce fiber for craft activities has been hindered by the lack of an
efficient plant propagation system. Seeds are highly unviable and cuttings need
large stem pieces to root (González, 1997).
This situation has increased pressure on natural populations as the major
source to obtain fiber for crafting. Reports show micropropagation throughout
culture of explants with pre-existing meristems as an efficient alternative
method for mass clonal propagation due to high multiplication rates, genetic
stability and complete plantlet recovery (Suárez, et al., 2009; Pastrana and Suarez, 2009;
Suárez, 2019b). Despite benefits of micropropagation for large
production of clonal plant material, costs of micropropagated plants are still
a disadvantage for low cost agricultural systems such as arrow cane cultivation. To lower costs in arrow cane
micropropagation, conventional semisolid culture media compared to double phase
medium system were evaluated; shoot costs were reduced by 40% when plants were
proliferated in double phase medium (López and Suárez, 2018). Likewise, ex vitro transfer and hardening of
micropropagated arrow cane plants without transparent plastic covers not only
reduced labor costs, but also increased plant growth without affecting plant
survival (Pico, 2019). Transplant to ex
vitro conditions is the most critical stage for plant survival in
micropropagation (Suárez, et al.,
2020). Ex vitro acclimatization of
arrow cane micropropagated plants has traditionally been done by transferring in vitro grown plants into high cost
peat substrate because of high water retention that contributes to full plant
recovery (Suárez, et al., 2013; Suárez,
et al., 2020). Substrate mixes are
intended to provide support to the plants, retain moisture, allow drainage and
provide nutrients, especially for root growth and development; however, using
locally available materials such as sand, crops residues, compost, rice husk
and soil contribute to lower substrate associated costs (Dias, et al., 2018; Pascual, et al., 2018; Waman, et al. 2019). In the present research,
the effect of peat and two substrate mixes on plant recovery and growth were
evaluated and cost associated with the ex
vitro acclimatization stage calculated.
MATERIALS AND METHODS
Plant material
Plant material was obtained from in vitro maintained Cv “Criolla” plants during a year with monthly transfers to fresh medium. Explants consisted of three-stem clusters established in semisolid MS (Murashige and Skoog, 1962) medium with (in mg L-1) BAP (Benzilaminopurine) (0.5), myoinositol (100), sucrose (30000), thiamine HCl (0.4) and solidified with TC Agar (7000). Medium pH was adjusted to 5.7-5.8 with HCl or KOH previous to agar addition. The medium was sterilized by autoclave at 120 °C and 1.2 psi. A single explant was established per 125 cm3 borosilicate flask with 30 mL medium aliquots. Flasks were covered with two layers of heavy-duty aluminum foil and sealed with Parafilm®. Cultures were stored at 25 °C with 12 hours photoperiod (40 μmol m-1 s-1) provided by white cool fluorescent lamps with transfers to fresh medium every four weeks.
Substrate preparation and evaluation
Three substrate mixes (volume:volume): peat, peat + rice husks (1:1) and peat + sand (1:1) were prepared and evaluated. Sand was obtained from river shores and washed with potable water followed by sterile deionized water. Peat was obtained from commercially available product (Pindstrup®) while rice husk and sand were locally obtained and disinfected by heat (180 °C) in an oven for 48 hours. Parts were mixed, moistened and covered with polyethylene during 48 hours before use. Substrate mixes were individually dispensed in 72-plug plastic trays 48 hours before plant transfer to ex vitro conditions.
Ex vitro transfer
In vitro cultivated Cv “Criolla” plant clusters maintained in multiplication medium during four weeks were removed from the flaks, the medium residue was washed out with sterile distilled water (Figure 1a) and clusters (3 cm long) were established in plugs previously filled with the respective substrate mix. In each plug, a cluster was placed and the substrate around it compacted (Figure 1b). Once transplanted, plants were sprayed with sterile distilled water and trays were covered with translucent plastic covers (Figure 1c). Trays were maintained in a shade house (50% Saram®) with three sprays a day using sterile distilled water. After three days, plastic covers were side moved to allow air exchange; and after five days, trays were uncovered and maintained with fog irrigation twice a day (9:00 AM and 4:00 PM) for 1 minute each.
 Figure 1. Shoot plant clusters of Gynerium sagitatum Aubl. Cv “Criolla”
plants removed from culture medium (1a), transplanted in substrate mix (1b) and
maintained during ex vitro acclimatization
(1c).
Figure 1. Shoot plant clusters of Gynerium sagitatum Aubl. Cv “Criolla”
plants removed from culture medium (1a), transplanted in substrate mix (1b) and
maintained during ex vitro acclimatization
(1c).
Research consisted of a one-way factorial experiment
where the effect of substrate mix (Three treatments) on plant survival and
growth was evaluated. Treatments were distributed with a complete randomized
block design were each tray was the block with three replicates per treatment
for a total of 648 experimental units. Eight weeks after the transplant, the
number of survived plants was registered and the survival percentage
calculated. For each treatment, 10 plants were randomly selected, plant height
data were registered, analyzed with ANOVA (α = 0.05) and means separated with
Tukey Test (α = 0.05). Estimation of substrate effect on plant cost was
calculated based on substrate value and amount used, and number of plants
recovered 8 weeks after the transplant.
RESULTS AND DISCUSSION
Transfer to ex vitro and plantlet recovery
Survival percentage under ex vitro conditions was 90% for plants transplanted in peat + sand mix, 85% for plants transplanted in peat and 60% for plants transplanted in peat + rice husk mix (Figure 2). ANOVA allowed detecting statistical differences (Pr< 0.0001) as a result of the treatments. The Tukey test on collected data showed that survival percentage of plant transplanted in trays filled with peat + sand mix was statistically higher than those transplanted in peat alone and peat + rice husk mix.
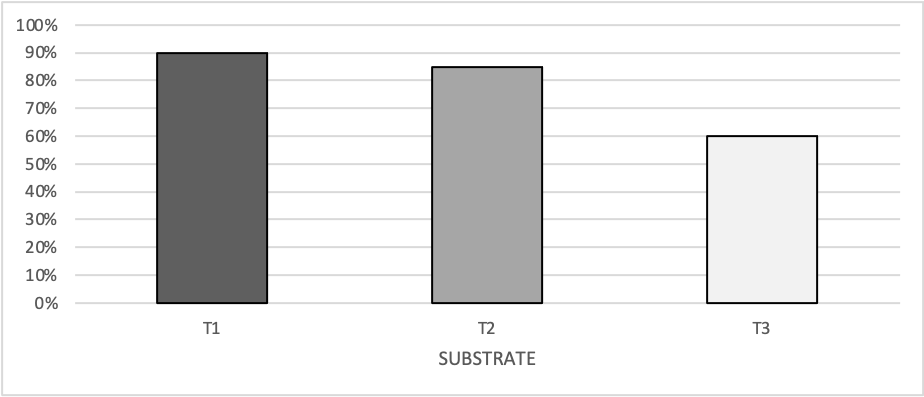 Figure 2. Percentage of Gynerium
sagitatum Aubl. Cv “Criolla” micropropagated
plants that survived after eight weeks of being transplanted in peat + sand mix
(T1), peat (T2) and peat + rice husk mix (T3) [Values with the same letter are not different according to Tukey test
(α = 0.05)].
Figure 2. Percentage of Gynerium
sagitatum Aubl. Cv “Criolla” micropropagated
plants that survived after eight weeks of being transplanted in peat + sand mix
(T1), peat (T2) and peat + rice husk mix (T3) [Values with the same letter are not different according to Tukey test
(α = 0.05)].
Plant height
The ANOVA allowed detecting statistical differences (Pr = 0.0042) in plant height data as a result of the effect of substrate mixes. Tukey mean separation test showed that plants transplanted in trays filled with peat + sand mix (9.18 cm) were significantly higher than the plants transplanted in peat + rice husk (8.43 cm) and those transplanted in peat alone (7.43 cm) (Figure 3).
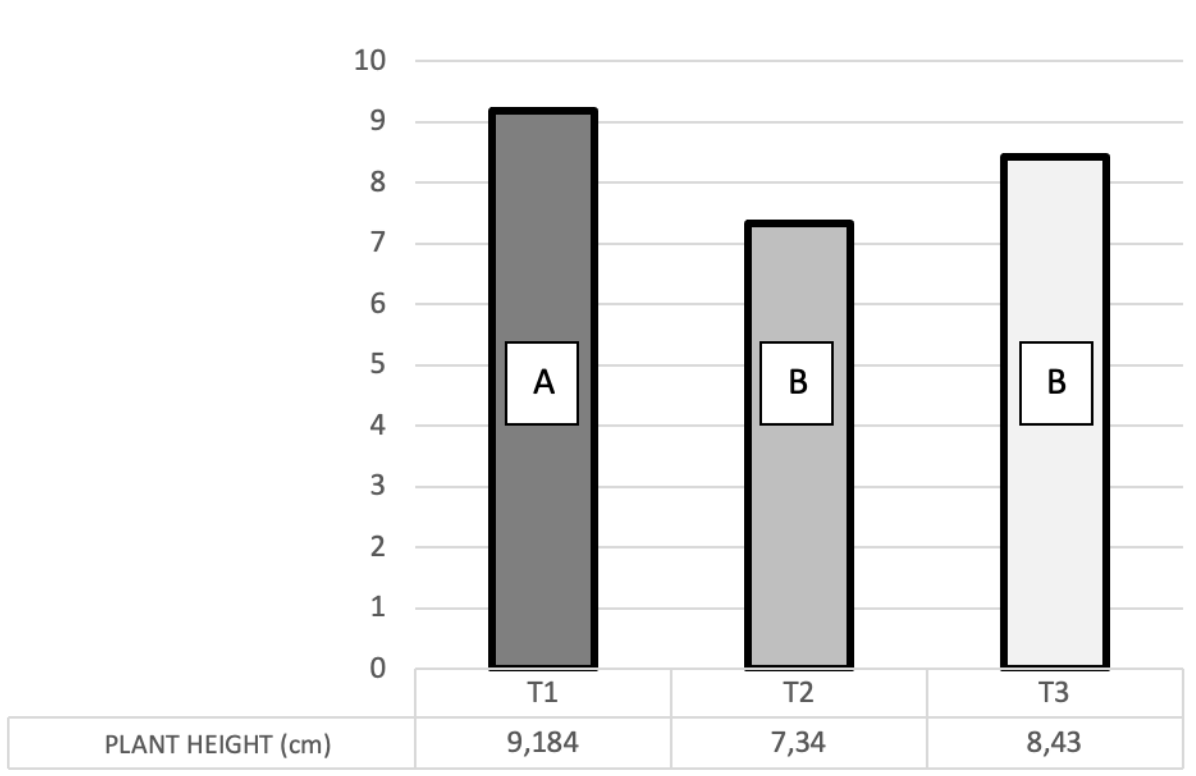 Figure 3. Plant height of Gynerium
sagitatum Aubl. Cv “Criolla” micropropagated
plants after eight weeks of being transplanted in peat + sand (T1), peat (T2)
and peat + rice husk (T3) [Values with
the same letter are not different according to Tukey test (α = 0.05)].
Figure 3. Plant height of Gynerium
sagitatum Aubl. Cv “Criolla” micropropagated
plants after eight weeks of being transplanted in peat + sand (T1), peat (T2)
and peat + rice husk (T3) [Values with
the same letter are not different according to Tukey test (α = 0.05)].
Cost analysis
The costs of substrate mixes used during plant transfer to ex vitro conditions varied from peat (US$5.52), peat + rice husk (US$4.14) and peat + sand (US$3.72), according with the amounts of materials used. The estimated cost of substrate mix associated with the total number of 216 transplanted plants in each substrate mix and the final number of plants recovered in each treatment, show that the highest economic efficiency in plant cost unit occurs when plants are transplanted in peat + sand mix, followed by peat alone and peat + rice husk with the highest proportional cost (Table 1).
Table 1. Cost analysis of ex vitro acclimatization of Gynerium sagitatum Aubl. Cv “Criolla” micropropagated plants transplanted in different substrate mixes.
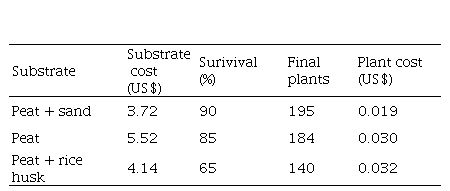
|
Substrate
|
Substrate cost
(US$)
|
Surivival
(%)
|
Final
plants
|
Plant cost
(US$)
|
|
Peat + sand
|
3.72
|
90
|
195
|
0.019
|
|
Peat
|
5.52
|
85
|
184
|
0.030
|
|
Peat + rice husk
|
4.14
|
65
|
140
|
0.032
|
Micropropagated plants are costly because of use of
specialized infrastructure, reagent costs, qualified personnel and labor (Ahloowalia and Savangikar, 2002; Aziz and Al-Taweel, 2019). Implementing
strategies to lower micropropagated plant costs is always a challenge, and studies
to increase cost efficiency in micropropagation of several plant species has
been reported (Raghu, et al., 2007;
Sahu and Kumar, 2013; George and Manuel, 2013; Kadam, et al., 2018). In arrow cane, mechanisms used to decrease plant
costs during micropropagation incorporate several aspects. López and Suárez
(2018) evidenced that using double phase (semisolid-liquid) culture media reduced
by 40% costs of Cv “Criolla” micropropagated
shoots and significantly increased in
vitro multiplication rate of Cvs “Criolla”,
“Criolla 1, “Martinera” and “Costera”
(López, 2018; Suárez, et al.,
2020). In a different strategy, Pico (2019) demonstrated that ex
vitro transplanting and hardening of micropropagated Cvs “Criolla”, “Martinera”
and “Costera” plants without plastic covers did not
affect survival rate and plant growth, but instead decreased costs because of
lower labor demand.
Several substrate mixes have been used to
increase survival during ex vitro
adaptation of various micropropagated plant species. Bonilla, et al., (2015) increased 3x the survival
rate and improved adaptation of micropropagated Manihot sculenta
plants when used a solid humus + rice husk (1:1) substrate mix.
Palacios-Arriaga, et al. (2019)
increased survival percentage of genetically modified rose plants regenerated
through somatic embryogenesis when the transplanting was done in a peat +
perlite (1:1) substrate mix. Espinosa-Reyes
et al. (2019) reported >85% survival and increased plant
height during ex vitro adaptation of Morus alba micropropagated
plants transplanted in substrate mixes with different amounts of soil, cow
manure and zeolite.
Sand is an inert material that provides
density, increase drainage and favors air exchange in substrate mixes; in
contrast, peat and rice husks favor water retention and moisture (Vheragen, 2009; Walczak, et al., 2002; Londra,
et al., 2018). Pérez-Alonso, et al. (2016) evaluated the effect of cachaza compost and zeolite, mixed or alone, on ex vitro adaptation of Aloe vera micropropagated
plants, observing that compost alone contributed to 100% plant survival and
better plant growth and development because of substrate aeration. Gil, et al. (2017) working on ex vitro transfer of micropropagated Morus alba plants reported an increased
percentage of ex vitro adapted
plants, higher number of rooted plants, increased mean number of leaves per
plant and larger leaves when plants were transferred in a sand + moss + humus
substrate mix, correlating the results with the sand characteristics that
provided to the mix. The present work evidenced that arrow cane micropropagated
plants showed a higher survival rate, a better growth and development, and
resulted in a lower costs when the transplanting was carried out in a peat + sand substrate mix. These results
allow increasing accessibility of the arrow cane micropropagation technology to
plant growers and artisans.
CONCLUSIONS
The peat + sand (1:1) substrate mix significantly increased plant survival in arrow cane Cv “Criolla” micropropagated plant transferred to ex vitro conditions.
Arrow cane Cv “Criolla” micropropagated plants transplanted to ex vitro conditions in a peat + sand (1:1) mix substrate grew significantly higher compared to plants transplanted in peat alone or peat + rice husk mix.
Ex vitro transfer of arrow cane Cv “Criolla” micropropagated plants using a peat + sand (1:1) substrate mix reduces plant costs associated to transplant by 35% with respect to plants transplanted in peat alone and by 37.6% when the transplant is done using peat + rice husk (1:1) substrate mix.
REFERENCES
Ahloowalia, B. and Savangikar, V. 2002. Low cost for energy and labor. In: Low cost options for tissue culture technology in developing countries. Proceedings of a Technical Meeting organized by the Joint FAO/IAEA Division of Nuclear Techniques in Food and Agriculture. Vienna August 2002, 26-30.
Artesanías de
Colombia. 2019. Artesanías de Colombia.
Colombia Artesanal. http://artesaniasdecolombia.com.co/PortalAC/General/template_index.jsf
[04 de junio 2019]
Aziz, R.A., and Al-Taweel, S. 2019. Effect of plant growth retardants on stevia (stevia rebaudiana bertoni) acclimatization produced in vitro. Plant Archives 19(1): 1275-1284.
Bonilla, M.M., Sánchez, S.A. and Pachón, Jorge. 2015. Evaluación de sustratos orgánicos para la aclimatación y endurecimiento de vitroplantas de yuca (Manihot esculenta Crantz). Revista de Investigación Agraria y Ambiental 6(2): 31-36p. 10.22490/21456453.1402.
Contreras, M., Espinosa, N. and Owen, M. 1998. Variabilidad del contenido de humedad en los periodos de luna llena y menguante, densidad y contracción del tallo de la caña brava. Gynerium sagittatum Aubl. Revista Forestal Venezolana 42: 97-102.
Dias, K., Pereira. L., Barbosa, M., Souza, V., Magalhães, P., Dos, E., Hurtado-Salazar, A. and Pereira, D. 2018. Development and root morphology of passion fruit in different substrates. Revista Colombiana de Ciencias Hortícolas 12(2):514-520p. 10.17584/rcch.2018v12i2.7779
Espinosa-Reyes, a., Silva-Pupo, J., Bahi-Arevich., M. and Romero-Cabrera., D. 2019. Influence of in vitro plant size and substrate type on the acclimatization
George, P. and Manuel, J. 2013. Low cost tissue culture technology for the regeneration of some economically important plants for developing countries. International Journal of Agriculture Environment & Biotechnology 6(4): 703-711.
González, O. 1997. Situación de dos métodos de siembra por estacas de caña flecha (Gynerium sagittatum Aubl.) de la variedad ´Martinera´ en la región de Montelibano, Córdoba. Tesis Ingeniero Agrónomo, Universidad de Córdoba, Monteria. 74 p.
Gil, A., López, S. and López, A. 2017. Aclimatación de plántulas in vitro de Saintpaulia ionantha H. Wendl. (Gesneriaceae) “violeta africana’’ a condiciones de invernadero. Arnaldoa 24 (1): 343 – 350p. 10.22497/arnaldoa.241.24116
GRIN (Germplasm Resources Information Network). 2019. [Online Database].National Germplasm Resources Laboratory, Beltsville, Maryland. [6 de marzo de 2019]
Kadam, D.D., Chhatre, A.A., Lavale, S.A. and Shinde, N.A. 2018. Low-Cost Alternatives for Conventional Tissue Culture Media. International Journal of Current Microbiology and Applied Sciences 7(4): 2523-2529p. 10.20546/ijcmas.2018.704.288.
Kalliola, R., Puhakka, M. and Salo, J. 1992. Intraspecific variation and the distribution and ecology of Gynerium sagittatum (Poaceae) in the western Amazon. Flora (Jena) 86: 153-167.
Londra, P., Paraskevopoulou, A. and Psychogiou, M. 2018. Hydrological Behavior of Peat- and Coir-Based Substrates and Their Effect on Begonia Growth. Water 10(6): 722p. 10.3390/w10060722
López, C. and Suarez, I. 2018. In vitro arrow cane (Gynerium sagitatum Aubl.) multiplication in double phase medium. Revista de Ciencias Agrícolas 35 (2): 5-13p. 10.22267/rcia.183502.86
López, C. 2018. Micropropagación in vitro de tres variedades de caña flecha (Gynerium sagitatum Aubl.) Criolla, Criolla 1 y Martinera. Tesis Maestría en Biotecnología, Universidad de Córdoba, Montería.
Murashige, T. and Skoog, F. A. 1962. Revised medium for rapid growth and bioassay with tobacco tissue culture. Physiologia Plantarum 15(3): 473.
Palacios-Arriaga, A. and Prasad, N. 2019. Efecto de diferentes medios y sustratos en el enraizamiento y adaptación de rosa transgénica. Agronomía Mesoamerica 30(1):115-129p. 10.15517/am.v30i1.29541
Pascual, J., Ceglie, F., Tuzel, Y., Koller, M., Koren, A., Hitchings, R. and Tittarelli, F. 2018. Organic substrate for transplant production in organic nurseries. A review. Agronomy for Sustainable Development 38 (35): 1-23. 10.1007/s13593-018-0508-4
Pastrana, I. and Suarez, I. 2009. Producción de plantas de caña flecha (Gynerium sagittatum) 'Criolla' a través de micropropagación. Revista Temas Agrarios 14 (2): 4-13p. 10.21897/rta.v14i2.671
Pérez-Alonso, N., Capote, A., Pérez, A., Gómez, L. and Chong-Pérez, B. 2016. Efecto del sustrato en la aclimatización de plantas in vitro de Aloe vera L. Biotecnología Vegetal 16(3): 161-169
Pico, D. 2019. Aclimatación ex vitro de plantas micropropagadas de tres variedades (“Criolla”, “Martinera” y “Costera”) de caña flecha (Gynerium sagitatum Aubl.). Tesis Ingeniero Agrónomo, Universidad de Córdoba, Monteria. 42p.
Raghu, A.V., Gerald Martin, V. and Priya, S.P. 2007. Low Cost Alternatives for the Micropagation of Centella asiatica. Journal of Plant Sciences 2(6): 592-599. 10.3923/jps.2007.592.599
Sahu, J. and Kumar, R. 2013. A Review on Low Cost Methods for In Vitro Micropropagation of Plant Through Tissue Culture Technique. Journal of Pharmaceutical and Biosciences 1(1): 38-41p.10.20510/ukjpb/1/i1/91115
Suárez, I., Aramendiz, H. and Pastrana, I. 2009. Micropropagación de Caña Flecha (Gynerium sagittatum Aubl.). Revista de la Facultad Nacional de Agronomía 62(2): 5135-5143p. 10.14198/cdbio.2011.37.01
Suárez, I., Pastrana, I. and Rivera, H. 2013. Biotecnología Aplicada a la Caña Flecha (Gyneriumsagitatum Aubl.). Fondo Editorial Universidad de Córdoba, Montería, Colombia, p200.
Suárez, I. 2019a. Biotecnología Aplicada a la Caña Flecha. Memorias LVI Congreso Nacional y V Internacional de Ciencias Biológicas. Sociedad Colombiana de Ciencias Biológicas. Montería, mayo de 2019, p20-24.
Suarez, I. 2019b. Micropropagación de tres variedades de caña flecha (Gynerium sagitatum Aubl.) en medio doble fase. Memorias XVIII Congreso Nacional de Biotecnología y Bioingeniería. Sociedad Mexicana de Bioingeniería y Biotecnología. Guanajuato, 23-29 de junio.
Suarez, I., Pérez, C. and López, C. 2020. Micropropagación de Gynerium sagitatum Aubl. cvs "criolla,"costera" y "martinera". Revista Biotecnología en el Sector Agropecuario y Agroindustrial 18 (2):60-69. http://dx.doi.org/10.18684/bsaa(18)60-69
Verhagen, J. 2009. Stability of growing media from a physical, chemical and biological perspective. Acta Horticulturae 819: 135-142p. 10.17660/ActaHortic.2009.819.12
Walczak, R., Rovdan, E. and Witkowska-Walczak, B. 2002. Water retention characteristics of peat and sand mixtures. International Agrophysics 16(2): 161-165.
Waman, A.A., Smitha, G.R. and Bohra, P. 2019. A Review on Clonal Propagation of Medicinal and Aromatic Plants through Stem Cuttings for Promoting their Cultivation and Conservation. Current agriculture Research Journal 7(2): 122-138p. 10.12944/CARJ.7.2.01









