



Dynamics of aerial and perennial biomass of two lucerne cultivars with different fall dormancy subject to two severities of cutting during the establishment phase
Dinámica de la biomasa aérea y perenne de dos cultivares de alfalfa con diferente grado de latencia invernal sujetos a dos severidades de corte durante la etapa de implantación
Revista de la Facultad de Agronomía
Universidad Nacional de La Plata, Argentina
ISSN: 1669-9513
Periodicity: Semestral
vol. 120, no. 1, 2021
Received: 20/07/2020
Accepted: 19/10/2020
Corresponding author: garcia.leonela@inta.gob.ar
Abstract: The aim of this study was to quantify the impact of two cutting severities on the dynamics of aerial and perennial (root plus crown) biomass of lucerne (Medicago sativa L.) plants of two cultivars of contrasting fall dormancy rating during the establishment phase. We worked at Unidad Integrada Balcarce with two cultivars of lucerne (non-winter dormant: FD10; semi-winter dormant: FD6) and two cutting severities (40 and 100 mm) defining four treatments (FD6-40 mm, FD6-100 mm, FD10-40 mm and FD10-100 mm) under a completely randomized design. In both cultivars, increasing cutting severity produced a reduction in leaf area and number of nodes in the residual, resulting in a reduced production of aerial biomass during regrowth, given a reduction on shoot number. On the other hand, the effect of increased cutting severity on perennial biomass was only observed in plants of FD10 cultivar. This cultivar showed less perennial biomass when cut at 40 mm than at 100 mm, while no differences were observed between cutting heights in FD6 cultivar.
Keywords: Medicago sativa, fall dormancy, cutting height, aerial biomass, root biomass.
Resumen: El objetivo de este estudio fue cuantificar el impacto de dos severidades de corte sobre la dinámica de la biomasa aérea y perenne (raíz más corona) de plantas de alfalfa (Medicago sativa L.) de dos cultivares que contrastan en su latencia invernal durante la implantación. Se trabajó en la Unidad Integrada Balcarce con dos cultivares de alfalfa (sin reposo invernal: FD10; con reposo invernal intermedio: FD6) y dos alturas de corte (40 y 100 mm) quedando definidos cuatro tratamientos (FD6-40mm, FD6-100mm, FD10-40mm y FD10-100mm) bajo un diseño completamente aleatorizado. En ambos cultivares, el incremento en la severidad de corte produjo una reducción en el área foliar residual y en el número de nudos de dicho residual lo que derivó en una reducción en la producción de biomasa aérea durante el rebrote, explicada por una reducción en el número de tallos. Por su parte, el efecto del incremento en la severidad de corte sobre la biomasa perenne sólo se observó en las plantas del cultivar FD10 que mostraron menor biomasa perenne cuando fueron cortadas a 40 mm de altura respecto de aquellas cortadas a 100 mm de altura, mientras que no se observaron diferencias entre alturas de corte para FD6.
Palabras clave: Medicago sativa, grado de reposo, altura de corte, biomasa aérea, biomasa raíz.
INTRODUCTION
The establishment phase of lucerne (Medicago sativa L.) refers to a period in which biomass is mainly partitioned to crown and roots (i.e. perennial biomass). This phase ends, independently of ontogeny, when crops reach an accumulation of perennial biomass of ∼5 t DM ha-1 (Sim et al., 2015). Understanding the responses of plants to defoliation during the establishment phase is important to develop optimal management practices, enabling increasing productivity and persistence of lucerne pastures (Sim et al., 2015).
Defoliations during the establishment phase are incompatible with the fast development of a vigorous root system (Moot et al., 2003; Thiébeau et al., 2011). However, on certain occasions, defoliation in the establishment phase of lucerne is inevitable due to food scarcity or to the excessive growth of other species competing for resources (e.g. weeds, accompanying grasses in forage mixtures). Grazing or cutting during the establishment phase would prolong the duration of this phase, since the removal of leaf area reduces the capture of radiation and generates a negative carbon balance, between inputs (via photosynthesis) and outputs (via respiration) of carbohydrates in the perennial biomass of plants (Teuber et al., 1998). In addition, the mobilization of nitrogen and carbon reserves stored in the perennial biomass to sustain the post-cutting regrowth, contributes to the reduction of root biomass (Avice et al., 1996). This effect could be augmented in case of highly severe defoliations (e.g. 0-5 cm of cutting height) where the low leaf area residual post-cutting increases the negative carbon balance (Avice et al., 1996). The negative impact of defoliation could be mitigated by leaving a greater amount of foliar residual (Langer & Steinke, 1965; Meuriot et al., 2005; Favero et al., 2008).
Lucerne cultivars are grouped according to their fall dormancy or winter growth activity (FD). Non-dormant cultivars (e.g. FD10) are usually more productive during autumn-winter, less tolerant to freezing injury, taller and with longer internodes than semi-dormant cultivars (e.g. FD6) (Thiebeau et al., 2011). Previous mentioned differences suggest that, at a similar cutting height, the negative impact of defoliation will be lower in a semi-dormant cultivar because the shorter internode length will leave a higher source (i.e. leaf area residual) and sink (i.e. nodes and internodes) capacity for regrowth than in a winter-active cultivar.
Despite the numerous studies carried out in lucerne few works focus on the management of defoliation at the establishment phase. The aim of the present work was to analyze the response, to two cutting severities during the early establishment phase in two lucerne cultivars contrasting in their winter activity.
MATERIALS AND METHODS
Site and experimental conditions
The experiment was carried out in a glasshouse and outdoors at Balcarce, Argentina (37° 45’ S, 58° 18’ W), under natural photoperiod from September 2015 to January 2016. Two cultivars were studied: the semi-dormant WL611 (FD6) and the non-dormant WL2058 (FD10). On 29 September 2015 polyvinyl chloride (PVC) containers of 780 mm height and 160 mm diameter, lined with polystyrene bag perforated at the bottom were filled with a uniform mixture of topsoil from a typical Argiudol (6% OM; 33 ppm P Bray 1) and river sand (1:1 v/v) and 10 seeds of the corresponding cultivar were sown in each container. Prior to sowing, all containers were irrigated to saturation and afterwards were watered every two days with tap water in order to maintain soil water content close to field capacity. The experiment was fertilized three times (30 October 2015; 15 December 2015; 5 January 2016) applying 100 kg N ha-1 as urea and a 20 kg P ha-1 as phosphorus pentoxide each time; containers were hand-weeded and chemical control of insects (Pantomorus leucoloma and Trips sp.) was performed on three occasions (22 December 2015; 4 January 2015 and 13 January 2015) using 0.3 L ha-1 of Chlorpyrifos (48%). On 19 October 2015 the seedlings were thinned leaving 2 plants per container. The containers were accommodated in a glasshouse to simulate a 500,000 plants ha-1 pasture. An external row of spare containers was added. On 11 November 2015 containers were moved outside the greenhouse maintaining the original design and on 15 December 2015 all plants were cut at 50 mm height. The first destructive sampling (H0) was carried out on 5 January 2016 and two cutting height treatments (40 mm and 100 mm from soil level) were applied. During the regrowth period (350°Cd, base temperature 5°C), three successive cuts at the corresponding heights were performed at approximately every 115°Cd (H1: 12 January 2016; H2: 19 January 2016 y H3: 26 January 2016). On each cutting date 4 containers per treatment were harvested. The harvested material was stored at 5°C until further processing in the laboratory.
Climate data were obtained from the Meteorological Station of the INTA EEA Balcarce located 300 m from the experimental site. Mean daily average temperature was slightly higher than the historical value for the same period (22.3°C vs. 21.0°C, respectively), while the value of the mean daily incident solar radiation was similar to the historical value (22.2 Mj m-2).
Measurements and calculations
On each cutting date, the following measurements were carried out on harvested plants:
Plant height, biomass per plant and its components: previous to cutting, the heights of all plants were measured. After cutting, biomass between the upper level of the crown and the cutting level, 40 mm or 100 mm, depending on the cutting height (remaining biomass) was separated from the crown. Afterwards the crown and the 300 mm upper section of the tap root, which represent around 70% of the total biomass of the root system, were carefully washed with tap water and the crown was separated from the root by visually determining the transition zone between root and crown. Both, the remaining biomass and the above cutting level biomass were separated in leaf (blades + petioles) and stems. All fractions were kept at -22°C for 24 hours and then dried for 72 hours in a forced-air oven at 65°C to obtain dry mass. Perennial biomass was calculated as the sum of crown and root biomass, aerial biomass as the sum of leaf and stem, and total biomass as the sum of perennial and aerial biomass.
Biomass partitioning: the shoot proportion [(leaf + stem) / (leaf + stem + crown + root)], the perennial proportion [(crown + root) / (leaf + stem + crown + root)] and the root proportion in perennial biomass [root / (crown + root)], were calculated. For the first harvest (H0) only the leaf [leaf / (leaf + stem] proportion was calculated.
Number of shoots per plant: the number of shoots above the cutting height was recorded.
Mean weight of shoots: the mean weight of shoots was calculated by dividing the aerial biomass (leaf + stem) by the number of shoots in the aerial biomass.
Origin and type of shoots in the remaining biomass: the shoots in the remaining biomass were classified as “crown shoots” or “axillary shoots” depending on whether they were originated directly from the crown or from axillary buds of crown shoots. Additionally, they were classified into “old” (rigid, hollow and yellow brownish‑colored) or “young” (flexible, solid and green‑colored).
Number of nodes in young shoots per plant: the number of nodes in each young shoot was recorded to calculate the total number of nodes per plant located in young shoots.
Leaf area of the remaining biomass per plant: A subsample of leaf blade of the remaining biomass was scanned at a resolution of 200 dpi to determine leaf surface area. The images obtained were analyzed using Fiji-ImageJ (Schindelin et al., 2012). The subsamples were dried in an oven at 60°C until constant weight to determine dry weight. Specific leaf area (SLA, m2 g-1) was calculated as the ratio between leaf surface area and dry biomass of the corresponding subsample. Leaf area per plant was calculated as the product of leaf biomass per plant by its corresponding SLA value.
Statistical analysis
Treatments were arranged as the factorial combination of two lucerne cultivars (FD6 and FD10), two cutting heights (40 mm and 100 mm from soil level) and four sampling dates (H0, H1, H2 and H3) in a completely randomized design with four replicates. The angular transformation was applied to the variables expressed in percentage (Steel & Torrie., 1980). For all the variables considered, the average of two plants per container was analyzed by ANOVA using InfoStat v. 2017 (Di Rienzo et al., 2017). Means were separated using LSD at the 5% significance level.
RESULTS
How were the plants at the time of treatment imposition (H0)?
Plants of FD6 cultivar were 36% heavier than FD10 ones at H0 (Table 1). This difference was also observed in aerial biomass (stem and leaf) and the perennial biomass (crown and root 0‑300 mm) components, although it was not significant in the case of the crown (Table 1). Unexpectedly, plants of FD6 cultivar tended to be taller (p=0.09) than FD10 ones (Table 1).
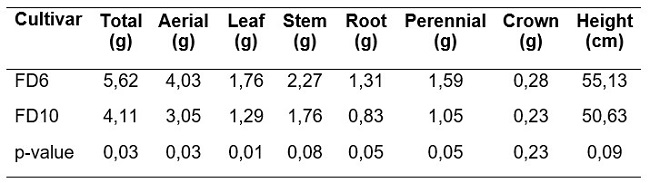
Both cultivars showed similar biomass partitioning at H0. Approximately 74% corresponded to aerial biomass and 26% to perennial biomass. Root biomass represented around 79% of the perennial biomass.
For both cultivars, after H0, plants cut at 100 mm had higher total biomass (+68%), leaf biomass (+320%), stem biomass (+53%) and number of nodes (+31%) in the residual, than those cut at 40 mm (Table 2). No differences (p>0.05) between cultivars were found for any of the analyzed variables.
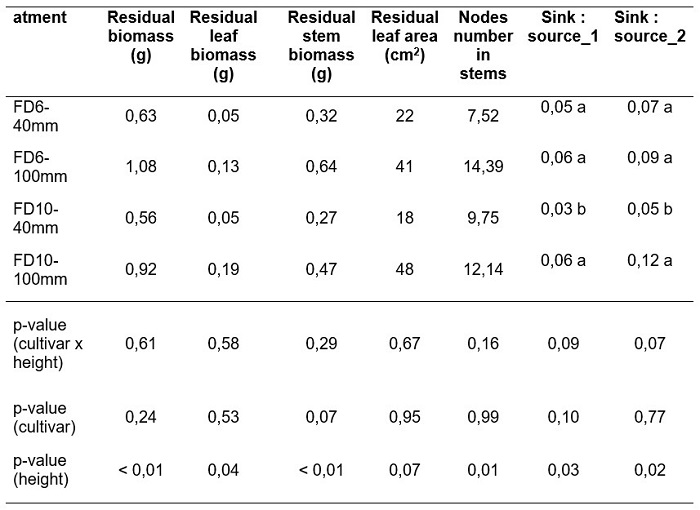
Two source/sink ratios were analyzed. The sum of leaf and green stems (young) biomass residual after the initial cutting (Table 2) was considered the source. The source was contrasted against two sinks. One sink was the number of nodes per plant in the residual (sink_1). The other sink was the number of young stems per plant in the residual (sink_2). Both source/sink ratios (source/sink_1 and source/sink_2) were significantly lower in FD10 plants cut at 40 mm height (Table 2).
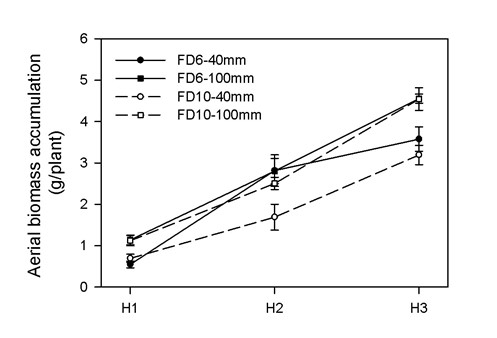
Dynamics of aerial biomass accumulation
Significant effects of cutting height and harvest date were detected (p<0.05). In general, for both cultivars plants cut at 100 mm showed higher aerial biomass accumulation than those cut at 40 mm (Figure 1).
Dynamics of the number and weight of shoots
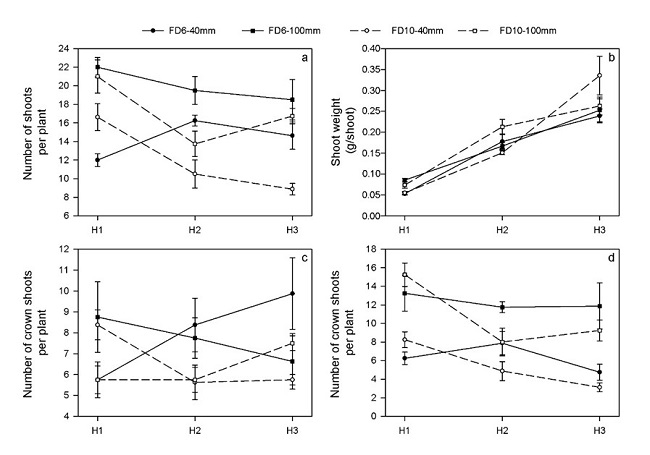
Total number of shoots per plant varied between 9 and 22 (Figure 2.a) and a significant cultivar x cutting height x harvest date interaction was found. Plants cut at 100 mm showed higher number of shoots than those cut at 40 mm, regardless of the cultivar and harvest date (Figure 2.a). At H2 plants of FD6 cultivar had a higher number of shoots than those of FD10 cultivar.
Individual shoot biomass varied between 0.05 and 0.35 g DM shoot-1 (Figure 2.b) and a significant interaction cultivar x cutting height x harvest date was found. While at the beginning of the regrowth, shoots were heavier in plants cut at 100 mm than at 40 mm, by the end of the regrowth shoots of FD10 plants cut at 40 mm were the heaviest (Figure 2.b). Moreover, while no differences between cultivars were found at the beginning of the regrowth, FD10 shoots were heavier than FD6 in H3.
The number of crown shoots per plant fluctuated between 6 and 10 (Figure 2.c) and a significant cultivar x cutting height x harvest date interaction was found. While at the beginning of the regrowth, FD6 plants cut at 100 mm showed a greater amount of crown shoots than those cut at 40 mm, the reverse was observed by the end of the regrowth period (Figure 2.c). The opposite response was found in FD10 plants (Figure 2.c).
The number of axillary shoots per plant fluctuated between 3 and 16 (Figure 2.d) and a significant interaction cultivar x harvest date was found. Regardless of the harvest date and cultivar evaluated, plants cut at 100 mm had higher number of axillary shoots than plants cut at 40 mm (Figure 2.d). Plants of FD6 cultivar had higher number of axillary shoots than FD10 at H2.
Dynamics of perennial biomass (crown + root)
Crown and taproot dry matter were linearly related (R2 = 0.80). The mass of both organs responded in a similar direction to management factors, and therefore crown and taproot dry matter were summed and analyzed as a single variable, the perennial biomass. The root mass was the major component (~ 80%) of the perennial biomass (data not shown).
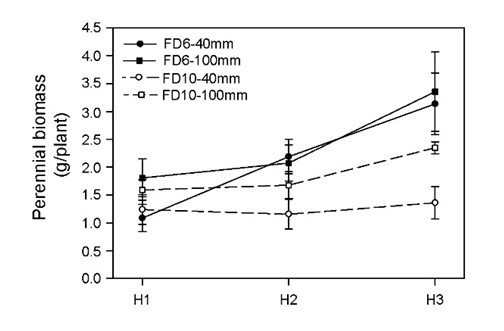
Total perennial biomass presented a significant interaction cultivar x harvest date and cutting height effect (Figure 3). Plants of FD6 cultivar showed a higher increase in perennial biomass during the regrowth than FD10 for which no differences among harvests dates were found (Figure 3). By the end of the regrowth period perennial biomass of plants cut at 100 mm were heavier than those of plants cut at 40 mm and this response was more evident in FD10 (Figure 3).
DISCUSSION
In this work, we examined the impact of cutting height on the behavior of two cultivars of lucerne during the early establisment phase. As in previous works (Hildebrand & Harrison, 1939; Langer & Steinke, 1965; Leach, 1969), plants less severely cut (100 mm) showed higher production of aerial biomass than those more severely cut (40 mm), this behavior being similar in both cultivars evaluated. The difference may be due to the greater amount of photosynthetic source (i.e. biomass of leaves, young stems and remnant leaf area) in plants cut at 100 mm of height. In this regard, numerous studies have shown the positive relationship between remaining green biomass and subsequent aerial growth in lucerne plants (Leach, 1969; Hodgkinson, 1973; Simon et al., 2004; Meuriot et al., 2005; Favero et al., 2008).
It is common to analyze aerial productivity of lucerne based on two components of yield: the number of shoots per plant and the individual shoot weight. In coincience with previous works (Leach, 1968; Leach,1970; Hodgkinson et al., 1972), differences between cutting height treatments were explained mainly by differences in the number of shoots per plant (Figure 2.a) since differences in shoot weight were minimal (Figure 2.b).
In lucerne, the aerial regrowth is derived from two types of shoots, those originated from the buds of the crown (crown shoots) and those originated from the axillary buds located in the nodes of the remaining stems (axillary shoots). In our work, the higher number of stems in plants cut at 100 mm (Figure 2.a) was explained by the axillary shoots (Figure 2.d). In both cultivars, the highest number of axillary shoots produced during the regrowth in plants cut at 100 mm, coincides with a higher number of nodes in the young stems of the post-cutting residual (Table 2).
As discussed above, it can be concluded that for the evaluated lucerne cultivars, the higher aerial growth of the plants less severely cut was explained not only by a greater amount of remaining photosynthetic source (i.e. greater residual leaf area), but also by a greater number of available sinks for the post-cutting regrowth (i.e. more nodes in young stems).
In contrast to what was observed in the aerial biomass, cutting height caused different responses between cultivars in the perennial biomass (crown + root). Plants of FD6 cultivar cut at 40 mm had lower perennial biomass than those cut at 100 mm at the beginning of the regrowth, but this difference disappeared afterwards (Figure 3). On the other hand, the plants of FD10 cultivar cut at 40 mm showed lower perennial biomass than those cut at 100 mm throughout the regrowth (Figure 3).
Why did FD10 showed reductions in aerial and perennial biomass in response to more severe cuts, while FD6 showed reductions only in aerial biomass? It has been observed that leaving more post-cutting remaining leaf area (and remaining green biomass) produces a lower dependence on the use of crown and root (perennial biomass) reserves for regrowth, especially during the first 15-21 days post-cutting (Avice et al., 1996; Meuriot et al., 2005). Thus, the lower perennial biomass observed in FD10 plants cut more severely could be explained by the lower availability of remaining leaf area when cut at 40 mm. Nevertheless, in the present work both cultivars were similarly affected by cutting heights showing similar post-cutting remaining leaf area, leaf biomass and young stem biomass (green) (Table 2). A possible explanation, not explored here, is that the residual leaves and stems of FD6 plants had greater photosynthetic capacity than those of FD10 plants, especially in plants cut at 40 mm height.
Another possible explanation is a different source/sink ratio between treatments in residual tissues after cutting. The presence of remaining green biomass reduces the use of carbon and nitrogen stored in the perennial biomass during the first days of the regrowth (Meuriot et al., 2005). The fact that both source/sink ratios were significantly lower in FD10 plants cut at 40 mm height (Table 2) suggests that, during the evaluated regrowth period they had lower source per unit of aerial sink. This could derive in a greater use of reserves for aerial growth and/or a diminished growth of perennial biomass in FD10 plants cut at 40 mm. Conversely, FD6 plants did not show differences in perennial biomass between cutting heights because under both cutting heights, 40 mm and 100 mm, the source/sink ratio at the beginning of the regrowth was similar.
It is important to note that at the beginning of the experiment, FD6 plants were larger than FD10 ones (Table 1), which coincides with the general observation that cultivars with greater fall dormancy have larger roots and crowns (Rodríguez & Spada, 2007). If these differences in plant size are the origin of the different responses of FD6 and FD10 in perennial biomass mentioned above is something that merit further research.
CONCLUSIONS
This study showed that, for both cultivars studied, the increment in the severity of cutting produced a similar reduction in aerial biomass. In turn, a different response was observed for perennial biomass, since only the non-dormant cultivar (FD10) was negatively affected by the increment in cutting severity.
BIBLIOGRAFY
Avice, J.C., A. Ourry, G. Lemaire & J. Boucaud. 1996. Nitrogen and carbon flows estimated by 15N and 13C pulse-chase labelling during regrowth of alfalfa. Plant Physiology 112: 281-290.
Di Rienzo, J.A., F. Casanoves, M.G. Balzarini, L. Gonzalez, M. Tablada & C.W. Robledo. 2017. InfoStat versión 2017. Universidad Nacional de Córdoba. [en línea].http://www.infostat.com. Último acceso: febrero de 2017.
Favero, D., S.M. Scheffer-Basso, M. Dall'Agnol & D. Seco. 2008. Desempenho de populações de alfafa sob desfolhação. Revista Brasileira de Zootecnia 37(4): 589-595.
Hildebrand, S.C. & C.M. Harrison. 1939. The effect of height and frequency of cutting alfalfa up on consequent top growth and root development. Journal of the American Society of Agronomy 31: 790-799.
Hodgkinson, K.C. 1973. Establishment and growth of shoots following low and high cutting of lucerne in relation to the pattern of nutrient uptake. Australian Journal of Agricultural Research 24: 497-510.
Hodgkinson, K.C., N.G. Smith & G.E. Miles. 1972. The photosynthetic capacity of stubble leaves and their contribution to growth of the lucerne plant after high level cutting. Australian Journal of Agricultural Research 23: 225-238.
Langer, R.H.M. & T.D. Steinke. 1965. Growth of lucerne in response to height and frequency of defoliation. Journal Agricultural Science 69: 291-294.
Leach, G.J. 1968. The growth of the lucerne plant after cutting: the effects of cutting at different stages of maturity and at different intensities. Australian Journal of Agricultural Research 19: 517-530.
Leach, G.J. 1969. Shoot number, shoot size and yield of regrowth in three lucerne cultivars. Australian Journal of Agricultural Research 20: 425-434.
Leach, G.J. 1970. Shoot growth on lucerne plants cut at different heights. Australian Journal of Agricultural Research 21: 583-591.
Meuriot, F., M.L. Decau, A. Morvan-Bertrand, M.P. Pru'Home, F. Gastal, J.C. Simon, J. Volonec & J.C. Avice. 2005. Contribution of initial C and N reserves in Medicago sativa recovering from defoliation: impact of cutting height and residual leaf area. Functional Plant Biology 32: 321-334.
Moot, D.J., H.E. Brown, E.I. Teixeira & K.M. Pollock. 2003. Crop growth and development affect seasonal priorities for lucerne management. In: Legumes for Dryland Pastures. Moot, D.J., Ed. New Zealand Grassland Association, Lincoln University, Christchurch, New Zealand, pp. 201–208.
Rodríguez, N. & M. del C. Spada. 2007. Morfología de la alfalfa. En: El Cultivo de la alfalfa en la Argentina. Basigalup D.H., Ediciones INTA, Buenos Aires, pp. 27-44.
Schindelin, J., I. Arganda-Carreras, E. Frise, V. Kayning, M. Longair, T. Pietzch, S. Preibisch, C. Rueden, S. Saalfeld, B. Schmid, J. Tinevez, D. White, V. Hartenstein, K. Eliceiri, P. Tomancak & A. Cardona. 2012. Fiji: an open-source platform for biological-image analysis. Nature methods 9: 676-682.
Sim, R.E., D.J. Moot, H.E. Brown & E.I. Teixeira. 2015. Sowing date affected shoot and root biomass accumulation of lucerne during establishment and subsequent regrowth season. European Journal of Agronomy 68: 69-77.
Simon, J.C, M.L. Decau, J.C. Avice, A. Jacquet, F. Meuriot & J.M. Allirand. 2004. Effects of initial N reserve status and residual leaf area after cutting on leaf area and organ establishment during regrowth of alfalfa. Journal Plant Scientist 84: 1059-1066.
Steel, R.G.D. & J.H. Torrie. 1980. Principles and procedures of statistics. A biometrical approach. 2nd ed. McGraw Hill Book Company. 633 pp.
Teuber, L.R., K.L. Taggard, L.K. Gibbs, M.H. McCaslin, M.A. Peterson & D.K. Barnes. 1998. Fall dormancy. In: Standard Tests to Characterize Alfalfa Cultivars. 3rd ed. Fox, C., Berberet, R., Gray, F., Grau, C., Jessen, D., Peterson, M. Eds. North American Alfalfa Improvement Conference. Agronomic Test, p. A-1.
Thiebeau, P., N. Beaudoin, E. Justes, J.M. Allirand & P.A. Maron. 2011. Radiation use efficiency and root dry matter partitioning in seedling growths and regrowth crops of Lucerne (Medicago sativa L.) after spring and autumn sowings. European Journal of Agronomy 35: 255-268.
Author notes
garcia.leonela@inta.gob.ar

