



Artículos
Mixed probiotic/anti-mycotoxin additive (Saccharomyces cerevisiae RC016 and Lactobacillus rhamnosus RC007) supplementation of AFB1-contaminated feed influences broiler chickens productive parameters, biochemistry and liver/intestine histopathology
Aditivos mixtos de probióticos/anti-micotoxinas (Saccharomyces cerevisiae RC016 y Lactobacillus rhamnosus RC007) suplementados con alimentos contaminados con AFB1 y su influencia en los parámetros productivos, la bioquímica y la histopatología del hígado/intestino de pollos de engorde
Ab intus FAV-UNRC
Universidad Nacional de Río Cuarto, Argentina
ISSN-e: 2618-2734
Periodicity: Semestral
vol. 6, no. 12, 2023
Received: 26 December 2022
Accepted: 28 November 2023
Funding
Funding source: This study was supported by grants from SECyT-UNRC 331/16, PIP-CONICET GI 11220120100156 and PICT 3089/18.
Corresponding author: lcavaglieri@exa.unrc.edu.ar
Abstract: The objective of the present work was to study the influence of dietary supplementation of a probiotic and anti-mycotoxin mixed additive (MA, Saccharomyces cerevisiae RC016 and Lactobacillus rhamnosus RC007) and their interaction on the performance and health (biochemistry and livers/intestines histopathology) of broiler chickens fed aflatoxin B1 (AFB1) contaminated diets. A total of 60 one-day-old Cobb broilers were randomly allocated into four treatment groups with three replicates of 5 birds each for a five-week d feeding experiment. The dietary experimental of each treatment (T) were formulated as follows: T1, a commercial diet (CD); T2, CD + AFB1 (506.14 ± 22.1 ng/kg); T3, CD + 0.1% MA, the ratio of S. cerevisiae RC016 (1 x 107 cells/g) to L. rhamnosus RC007 (1 x 108 cells/g) was 1:1; T4, CD + AFB1 (506.14 ± 22.1 ng/kg) + 0.1% MA. The MA improved (p<0.01) production parameters (weight gain, conversion rate, and carcass yield) and reduced (p < 0.01) the toxic effect of AFB1 on the relative weight of the livers. In addition, the macro and microscopic alterations of livers and the possible intestinal injury related to histological damage in the presence of mycotoxin were reduced. The use of probiotic MA based on S. cerevisiae RC016 and L. rhamnosus RC007 in animal feed provides greater protection against mycotoxin contamination and is safe for use as a supplement in animal feed, exercising benefit effects that improve animal health and productivity. This is of great importance at the economic level for the avian production system.
Keywords: broiler chickens, probiotic, antibiotic, aflatoxin B., growth performance..
Resumen: El objetivo del presente trabajo fue estudiar la influencia de la suplementación dietética de un aditivo mixto de probióticos y anti-micotoxinas (MA, Saccharomycescerevisiae RC016 and Lactobacillus rhamnosus RC007) y su interacción sobre el rendimiento y la salud (bioquímica e histopatología de hígado/intestino) de pollos de engorde alimentados con una dieta contaminada con aflatoxina B1 (AFB1). Un total de 60 pollos de engorde Cobb de un día de edad se asignaron al azar en cuatro grupos de tratamiento con tres réplicas de 5 aves cada una durante cinco semanas. La dieta experimental de cada tratamiento (T) se formuló de la siguiente manera: T1, dieta comercial (CD); T2, CD + AFB1 (506.14 ± 22.1 ng/kg); T3, CD + 0.1% MA, la relación fue 1:1, S. cerevisiae RC016 (1x107 cells/g) a L. rhamnosus RC007 (1x108 cells/g); T4, CD + AFB1 (506.14 ± 22.1 ng/kg) + 0.1% MA. La MA mejoró (p<0.01) los parámetros de producción (ganancia de peso, tasa de conversión y rendimiento de la canal) y redujo (p < 0.01) el efecto tóxico de AFB1 sobre el peso relativo de los hígados. Además, se redujeron las alteraciones macro y microscópicas de los hígados y la posible lesión intestinal relacionada con el daño histológico en presencia de micotoxinas. El uso de probióticos MA a base de S. cerevisiae RC016 y L. rhamnosus RC007 en la alimentación animal proporciona una mayor protección contra la contaminación por micotoxinas y es seguro para su uso como suplemento en la alimentación animal, ejerciendo efectos beneficiosos que mejoran la salud y la productividad animal. Esto es de gran importancia a nivel económico para el sistema de producción aviar.
Palabras clave: pollos de engorde, probióticos, antibióticos, aflatoxina B., parámetros productivos..
INTRODUCTION
The poultry industry is one of the fast-growing sections among the different agricultural sectors. The need to increase the efficiency of productive systems, not only the production of meat but also their competitiveness, has given rise to an intensification process of productive systems in which different challenges have been presented such as stress, antibiotics and modern breeding practices. For several decades, animal growth promoting antibiotics (GPA) were used in sub-therapeutic doses as additives to improve animal health and well-being, as well as to increase growth, to improve meat production through increased food conversion, and disease prevention (Ronquillo and Hernandez 2017; Alagawany et al., 2018; Mehdi et al., 2018). The abusive use of GPA and the associated selection pressure decreased the therapeutic efficacy and created populations of antibiotic-resistant microorganisms. Due to the prohibition of the use of GPA, there is a growing demand for alternative additives that provide benefits for animal health and growth worldwide.
A wide variety of non-therapeutic alternatives that can replace antibiotics such as probiotics, prebiotics, enzymes, organic acids, immunostimulants, bacteriocins, bacteriophages, phytogenic additives, nanoparticles, and essential oils were considered (Park et al., 2016; Peng et al., 2016). Probiotics are gaining acceptance as alternatives to GPA to improve production efficiency. They are mono or mixed cultures of living organisms that when administered in adequate amounts confer a health benefit to the host (FAO/OMS, 2001). Probiotics can be administered alone or in combination with other additives in food or water. In addition, there are many studies that demonstrate their ability to interact with food contaminants and thus, reduce the amount that reach the bloodstream to reduce their bioavailability (Chiocchetti et al., 2019; Śliżewska et al., 2019; Arif et al., 2020). A variety of bacteria (Bacillus, Bifidobacterium, Enterococcus, Lactobacillus, Streptococcus and Lactococcus spp.) and yeasts (Saccharomyces spp.) were tested as probiotic additives in poultry. Their use should maintain good health and not affect the environment. Moreover, they should improve performance characteristics that include among others improved feed conversion ratio, average daily weight gain, egg production, the carcass composition, the reproductive performance of breeding females, and also decrease the incidence of diseases, exhibiting a growth-promoting effect (Blajman et al., 2015; Caly et al., 2015; Abd El-Moneim et al., 2020). The application of yeasts in feed, and the status as microorganisms generally recognized as safe (GRAS) make them an adequate basis for strategies designed to reduce oral exposure to chemical contaminants such as mycotoxins. The animal feed industry needs to produce high nutritional value and microbiological quality feeds, to ensure good animal health and performance, whereas replacing the use of GPA. The effective use of feed additives containing yeast strains and/or lactic bacteria (LB) in mixture as adsorbents and/or mycotoxin degraders with probiotic properties is a promising alternative. Based on the above, the objective of the present work was to study the influence of dietary supplementation of a probiotic and anti-mycotoxin mixed additive (MA, Saccharomyces cerevisiae RC016 and Lactobacillus rhamnosus RC007) and their interaction on the performance and health (biochemistry and livers/intestines histopathology) of broiler chickens fed aflatoxin B1 (AFB1) contaminated diets.
MATERIALS AND METHODS
The working protocol and the used techniques comply with the regulations of the Subcommittee on Animal Bioethics under the Ethics Committee of Scientific Research, as established in Resolution 253/10 of the Superior Council of the National University of Rio Cuarto.
Microorganisms
Saccharomyces cerevisiae RC016 was isolated from animal ecosystem and identified by molecular techniques through DNA extraction and 18S rRNA and 28S rRNA amplification and analysis, comparing sequences with the Basic Local Alignment Search Tool (BLAST) within the National Centre for Biotechnology Information (NCBI) database (Armando et al., 2012).
Lactobacillus rhamnosus RC007 was isolated from maize silage and identified from both the fermentation pattern (API 50 CHL test) and the 16S rRNA gene sequence (Dogi et al., 2013). These strains are deposited in the culture collection of the Industrial Microbiology Laboratory of the National University of Rio Cuarto collection centre, located in Río Cuarto, Córdoba, Argentina.
Yeast and bacteria biomass production and formulation
Saccharomyces cerevisiae RC016 biomass was obtained from 24 h culture in Yeast-Peptone-Dextrose (YPD) broth added 1 g PO4H2K/L in a BioFlo 2000 fermentor (New Brunswick Scientific Co., Inc, Enfield, CT, USA) operated at 4 x . at 28 ºC, for 12 h and 1.5 vvm aeration. The pH value was adjusted to 5 with 6 M NaOH. The working volume was 4 L.
Lactobacillus rhamnosus RC007 culture conditions were 3 L of optimized culture medium developed with a low-cost substrate (commercial refinery syrup), stirring 4 x g at 37 °C, for 24 h and 10% inoculum (v/v). The concentration of dissolved oxygen at the beginning of the experiment was 0%. Foam production was controlled by the addition of antifoam 289 (Sigma-Aldrich, St. Louis, MO, USA). The pH was maintained between 6.5-7, with the addition of 18 N H2SO4 or Na2CO3 20% w / v.
The biomass obtained at the end of the fermentation was centrifuged at 1000 x g at 4 °C for 10 min. The concentrated pellet was resuspended in the same volume of cryoprotectant (10% skim milk plus 5% yeast extract, for the yeast and 10% skim milk only for the bacteria) and stored at – 80 °C. The lyophilized formula (1 g) was hydrated and the viability was confirmed. Finally, the mixed additive (MA) formulation was made mixing the lyophilized microorganisms (1:1) as follows: S. cerevisiae RC016 at 1x107 cells/g and, L. rhamnosus RC007 at 1x108 cells/g and then, 0.1% MA (0.1 g MA per 100 g commercial diet -CD- described below) was used for the in vivo trial.
Aflatoxin B. production
Sufficient AFB1 was produced to contaminate the feed for the experiment according to the methodology proposed by González Pereyra et al. (2014) from the culture of the reference strain Aspergillus parasiticus NRRL2999. The AFB1 content of the resulting powder was quantified by high performance liquid chromatography (HPLC) according to the methodology described by Trucksess et al. (1994). Analyses were performed in triplicate. The AFB1 contaminated powder was then added to the premix to reach a final concentration of 506.14 ± 22.1 ng/kg. Diet without AFB1 addition had 22.15 ± 1.15 ng/kg natural AFB1 contamination.
Experimental design
Animals
This experimental study was repeated twice. One-day old male Cobb 500 (0.086 ± 0.020 kg, total n = 60) vaccinated against Marek's disease obtained from a commercial hatchery were used. The broiler chickens were acclimatised for a period of one week. Water and feed were provided ad libitum. They were then randomly housed in stainless steel metabolic cages (3 replicates of 5 animals each/cage). The broiler chickens were raised in cages instead of floor pens to reduce the operational cost. Each cage contained a tube feeder and drinker and was covered with fresh chips. Animals were kept under continuous fluorescent light. The temperature was adjusted daily to maximise the comfort of the birds.
Diets
The broiler chickens were fed a commercial starter diet from hatch until 28 d of age (4 weeks) and a commercial grower diet from 29 to 35 d of age (4 to 5 wks). The diet was corn and/or soy-based, which meets the rules and regulations of the National Research Council (NRC) requirements for broiler chickens (Dale, 1994). The chickens were fed with the experimental diets from 1 d until slaughter at 35 d of age (Table 1). The experimental diets for each treatment (T) were formulated as follows: T1: commercial diet (CD); T2: CD + AFB1 (506.14 ± 22.1 ng/kg); T3: CD + AFB1 (506.14 ± 22.1 ng/kg) + 0.1% MA (1:1) from lyophilized microorganisms: S. cerevisiae RC016 (1x107 cells/g) and L. rhamnosus RC007 (1x108 cells/g); T4: CD + 0.1% MA.
| Diets | Centesimal composition | Time | ||
| Starter | Proteins 20% min. Ethereal extract 5% min Calcium min/max: 1.0 - 1.2% Phosphorous min/max: 0.7 - 0.9% Crude fibre 3% max. Total minerals 10% max. Moisture 13% max. Digestible energy: 3.000 Kcal/Kg. | Free access from the first day of life to the fourth week (28 days) included. | ||
| Finisher | Proteins 18% min. Ethereal extract 4% min. Calcium min/max: 0.9 - 1.1% Phosphorous min/max: 0.7 - 0.9% Crude fibre 3% max. Total minerals and vitamins 10% max. Moisture 13% max. Digestible energy: 3100 Kcal/Kg. | From the fifth week of age (29 days) until reaching the market weight. | ||
Productive parameters determination
Broiler chickens were weighed weekly during the experimental period (five weeks) as well as feed intake was calculated. Also, animals were monitored daily for signs of morbidity and mortality. The efficacy of the MA was evaluated at the end of the feeding test by measuring the productive parameters Average Weight Gain (AWG) (g), Average Feed Intake (AFI), Feed Conversion Ratio (FCR) calculated by the relationship between AFI and AWG, during five weeks. Also, the weight and carcass yield were also determined according to the methodology proposed by Magnoli et al. (2017). Each of these growth parameters was measured individually (per animal) and per treatment, and statistically analysed.
Biochemical parameters
Blood samples (1 mL/bird) were collected at the end of the feeding test. The serum was then obtained by centrifugation (2,500 × g for 15 min at room temperature) and stored at -20 °C for the determination of total proteins (TP), albumin (Alb). The total fraction of globulin (Glob) was calculated subtracting the Alb from the TP, and the albumin:globulin ratio (Alb:Glob) ratio was determined. The enzymes aspartate aminotransferase (AST), alanine aminotransferase (ALT), gamma-glutamyltransferase (GGT) and lactate dehydrogenase (LDH) were also determined using the colorimetric method using a commercial kit (Wiener Lab).
Slaughter
After the fifth week, animals were slaughtered. Livers and intestines were collected. Liver weight was expressed as a percentage of the body weight (organ weight/100 g live body weight).
Liver and intestine histopathology
Portions of approximately 6 mm2 of liver and small intestine tissue samples (duodenum) were fixed in 4 % (v/v) buffered-saline formaldehyde pH 7.2-7.4 at 4 °C, dehydrated in a graded series of ethanol (30%, 50%, 70%, 80%, 90%, 95% and 100%) and xylene solutions, embedded in paraffin and cut in ± 4 µm histological serial-sections. The histological sections were stained with hematoxylin/eosin (H/E) for microscopic analysis. Liver slides were examined for characteristic intoxication signs and hepatocellular degeneration according to Magnoli et al. (2011). Intestines were examined for damage and inflammation using a standard histopathological grading system described by Del Carmen et al. (2013). High histological scores indicate increased damage in the intestines. Digital images were captured with an Axiophot microscope (Carl Zeiss, Thornwood, NY) fitted with a high-resolution Power shot G6 7.1 megapixels digital camera (Canon INC, Japan). Digital image analysis and morphometric measurements were performed with Axiovision AxioVs40 V4.6.3.0 software (Carl Zeiss, Göttingen, Germany).
Statistical analysis
Data were analysed by a general linear mixed model (GLMM) (version 2.03; Córdoba, Argentina). Data were analysed by analysis of variance (ANOVA). Means were compared using Fisher's protected least significant test (LSD) (p<0.05).
RESULTS
Productive parameters
Average weight gain, feed intake and feed conversion ratio determinations
The weekly average weight gain (AWG) for each treatment is shown in Table 2. During week 5, chickens that received only AFB1 (T2) significantly reduced the AWG (p<0.0155), whereas those that received the toxin and MA (T3) and MA (T4) reached a significant increase in AWG during week 4 and week 5 (p ≤ 0.0143 and p ≤ 0.0155) with values similar to the control treatment (T1). Table 2 shows the results obtained for FCR. Treatments with the best FCR were the treatment with toxins plus the MA (T3) and the mixed additive (T4), that is, a smaller amount of food was required for the production of a kilo of live weight, in comparison to the control (T1). In T2, the presence of AFB1 significantly reduced all the productive parameters tested (p <0.01).
| Treatments | Weight day 1 | Wk - 1 | Wk - 2 | Wk - 3 | Wk - 4 | Wk - 5 | ||||||||
| W | CR | W | CR | W | CR | W | CR | W | CR | |||||
| T1 | 86.8 a | 157.4b | 0.692 | 576.6b | 0.581 | 956.5b | 0.850 | 1259.7b | 1.177 | 1638.0c | 1.473 | |||
| T2 1 | 84.5 a | 147.4a | 0.644 | 533.3a | 0.553 | 904.9a | 0.782 | 1169.7a | 1.107 | 1334.1a | 1.728 | |||
| T3 2 | 87.8 a | 148.7a | 0.673 | 553.2a | 0.560 | 921.8ab | 0.799 | 1227.7b | 1.136 | 1351.9b | 1.732 | |||
| T4 | 89.4 a | 159.2b | 0.685 | 567.7b | 0.590 | 940.8b | 0.864 | 1246.4b | 1.190 | c | 1.487 | |||
| P – value | 0.001 | 0.0095 | 0.033 | 0.0021 | 0.026 | 0.0094 | 0.017 | 0.0143 | 0.050 | 0.0155 | 0.050 | |||
Productive parameters determination
Table 3 shows the performance of the carcass for each of the treatments tested. The treatment with MA (T3) and the combination of the toxin with MA (T4) had the highest values, and they were able to counteract the harmful effect of mycotoxin. The presence of AFB1 (T2) significantly reduced (p < 0.1743) the performance levels.
Liver weight and macroscopic lesions analyses
Table 3 shows the relative weights of the livers from the different treatments. A significant increase (p < 0.0131) was observed in the treatment with AFB1 (T2) compared to the other treatments. The addition of MA to the toxin diet decreased the toxic effect of AFB1 on the relative weight of livers.
| Treatments | Carcass (%) | Relative weight (%) |
| T1 - Commercial diet | 68.87 ± 10.48 bc | 1.982 ± 0.227 a |
| T2 - AFB1 diet1 | 63.99 ± 3.96 a | 2.423 ± 0.520 b |
| T3 - AFB1 + mixed additive diet 2 | 71.60 ± 2.73 c | 1.847 ± 0.092 a |
| T4 - mixed additive diet | 68.12 ± 0.86 abc | 1.991 ± 0.151 ab |
| P – value | 0. 1743 |
Biochemical parameters
To assess the health status of broiler chickens fed with the different treatments, the blood test was collected the day before slaughter. The levels of PT, ALB, GLOB, ALB: GLOB, and AST, ALT, GGT, LDH enzymes were determined (Table 4). Total proteins and ALB, in general, were similar among all the tested treatments. However, GLOB levels were significantly reduced in the presence of AFB1 (T2) (p≤0.05). This effect was reflected in the relationship between ALB and GLOB, significantly increasing this coefficient. There were no significant differences among treatments for all the enzyme values tested (p≥0.05).
| Treatments | PT (g/dL) | ALB (g/dL) | GLOB | ALB:GLOB | ALT (g/dL) | AST (g/dL) | LDH (g/dL) | GGT (g/dL) |
| T1 - Commercial diet | 0.99 ± 0.27 ab | 1.3 ± 0.4 b | 0.0543 | 17.22 | 1.35 ± 0.13 | 1.26 ± 0.03 | 2.59 ± 2.71 | 0.09 ± 0.05 |
| T2 - AFB1 diet1 | 0.96 ± 0.10 a | 1.1 ± 0.2 ab | 0.0266 | 35.14 | 1.43 ± 0.20 | 1.25 ± 0.05 | 1.25 ± 0.51 | 0.15 ± 0.06 |
| T3 - AFB1 + mixed additive diet 2 | 1.39 ± 0.44 b | 1.3 ± 0.3 b | 0.0463 | 28.96 | 1.47 ± 0.20 | 1.40 ± 0.18 | 0.89 ± 0.39 | 0.13 ± 0.05 |
| T4 - mixed additive diet | 1.14 ± 0.34 ab | 1.1 ± 0.2 ab | 0.0488 | 22.39 | 1.43 ± 0.15 | 1.32 ± 0.13 | 1.27 ± 0.28 | 0.11 ± 0.06 |
Figure 1 shows the macroscopic appearances of chicken livers that were fed the different experimental diets. In treatment 1 (T1) a liver of small size, smooth surface, bright and intense wine-red colour corresponding to a normal liver was observed. In animals fed a diet contaminated with AFB1 (T2), a lighter colour, friable and pale in appearance was observed compared to controls (T1). The diets containing MA (T3 and T4), showed similar appearance and colour as the control treatment (T1), suggesting the preventive effect in the presence of the toxin.

Livers of broiler chickens from the different treatment (T), T1 - Commercial diet (CD); T2 - CD + AFB1 (506.14 ± 22.1 ng/kg); T3 - CD + AFB1 (506.14 ± 22.1 ng/kg) + 0.1% mixed additive (MA), lyophilized microorganisms, the ratio of S. cerevisiae RC016 (1 x 107 cells/g) to L. rhamnosus RC007 (1 x 108 cells/g) was 1:1; T4 - CD + 0.1% MA.
Livers histopathology
Figure 2 shows the photomicrographs of livers stained with hematoxylin and eosin. In T1 and T4, the peripheral lobular (hepatic) aspect was observed slightly vacuolar (hydropic degeneration). The T1 livers (control) showed a uniform pink tone throughout the body and bile duct hyperplasia. Hepatocytes (from four contiguous histopathological sections) showed an empty cytoplasm with basophilic and contracted nuclei of different diameters. The interlobular space showed 2 to 3 bile ducts in the portal space and bloodless vessels. A typical picture of chronic mycotoxicosis was considered here (T1). The livers from animals receiving the MA showed hepatocytes with slight hydropic degeneration (vacuolar), considered a normal microscopic appearance (T4). At the peripheral lobular interstitial level, two bile ducts and the interlobular vein were seen. Liver of chickens fed the AFB1 contaminated diet (T2) showed a slight microvacuolar fatty degeneration of hepatocytes. The coronal lobular section had few sinusoidal spaces and little congestion. The cores were of regular size and shape. Liver of chickens fed the AFB1 contaminated diet and the MA (T3) had normal appearance and no proliferation of the bile ducts was observed. No microvacuolar fat degeneration was also observed, suggesting the protective effect of the MA in hepatic aflatoxicosis.
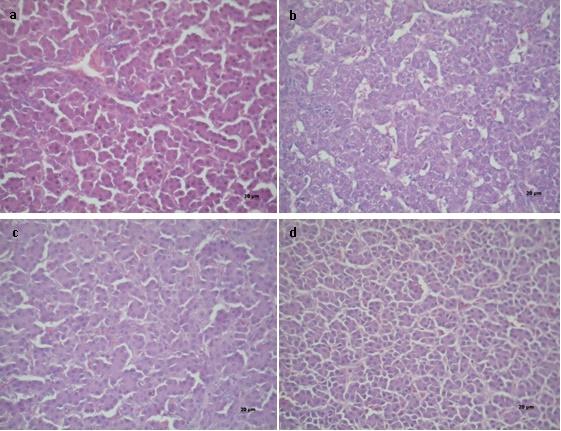
Histopathology of the livers of broiler chickens under different treatments (T), T1 - Commercial diet (CD); T2 - CD + AFB1 (506.14 ± 22.1 ng/kg); T3 - CD + AFB1 (506.14 ± 22.1 ng/kg) + 0.1% mixed additive (MA), lyophilized microorganisms, the ratio of S. cerevisiae RC016 (1 x 107 cells/g) to L. rhamnosus RC007 (1 x 108 cells/g) was 1:1; T4 - CD + 0.1% MA. 40X magnification.
Small intestines histological analyses
Figure 3 shows representative photomicrographs of the small intestine of each treatment at 2.5 x, 10 x and 40 x. The T1 treatment presented grade 0, according to the denomination established by Del Carmen et al. (2013) without evidence of epithelial alteration and the villi were intact. Likewise, the chickens that only received the MA (T4) were similar to the control treatment. The presence of AFB1 (T2) showed alterations, thickening of the submucosa of the small intestine (grade 1) and proliferation of the Brünner glands (indicated with arrow in Figure 3 40x). In contrast, T3 (AFB1+ MA) showed no histological damage in the small intestine, MA reduced or prevented a possible intestinal injury induced by AFB1.
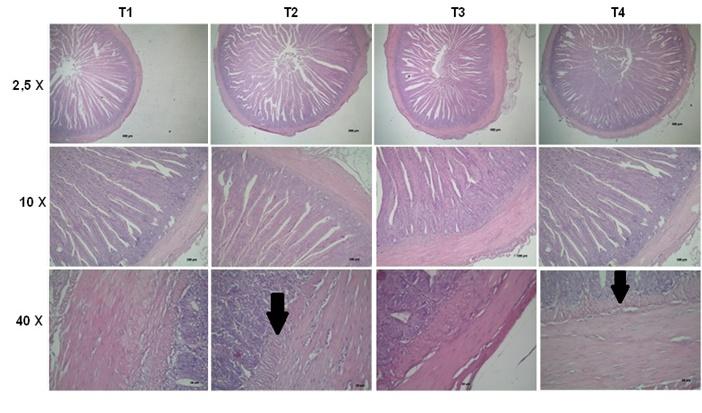
Representative microphotographs of the small intestine of each treatment (T), T1 - Commercial diet (CD); T2 - CD + AFB1 (506.14 ± 22.1 ng/kg); T3 - CD + AFB1 (506.14 ± 22.1 ng/kg) + 0.1% mixed additive (MA), lyophilized microorganisms, the ratio of S. cerevisiae RC016 (1 x 107 cells/g) to L. rhamnosus RC007 (1 x 108 cells/g) was 1:1; T4 - CD + 0.1% MA. arrow indicates the presence of Brünner glands.
DISCUSSION
Growth-promoting antibiotics are well documented to play an important role in the feed efficiency of poultry. Regulations and consumers demand decreased their use worldwide, resulting in increased research focused on the development of alternatives to maintain or improve poultry health and performance (Vuong, et al., 2016; Gadde, et al., 2017).
Saccharomyces cerevisiae RC016 and L. rhamnosus RC007 are strains proved industrially scalable that complement probiotic and anti-mycotoxin actions; in addition, they were demonstrated to be GRAS for use as supplements in animal feed. They also demonstrated improvement of animal health and productivity used in separate formulations (García et al., 2017; Fochesato et al., 2018,2019; Poloni et al., 2020).
The final application of the MA in the present work was carried out in an in vivo test with broiler chickens to assess its safety plus the probiotic and anti-mycotoxin potential in a challenge with AFB1. In general, the tested productive parameters (AWG, FCR, and carcass performance) improved significantly in treatments with the MA. These results coincide with those of Bai et al. (2013) with L. fermentum and S. cerevisiae as probiotics in different doses (0.1% to 0.3%) and two growing periods (1 to 21 d and 22 to 42 d) achieving better results at 0.1% product in the initial phase diets compared to antimicrobial growth promoters.
Here, the treatment that combined MA with the AFB1 diet managed to counteract the decrease in weight gain, carcass performance, and histopathological damage caused by aflatoxicosis. Similar results to ours were obtained by Śliżewska et al. (2019) that tested the efficacy of a probiotic preparation containing Lactobacillus and S. cerevisiae species and also reversed the negative effects of treatments experimentally contaminated with AFB1. However, they tested two different doses (1 and 5 mg/kg) and at a higher dose of AFB1, the probiotic could not avoid the histological changes induced by mycotoxin.
The determination of biochemical parameters allows to assess the nutritional status (Hasan et al., 2015). The liver has an essential role in the metabolism of nutrients, in the detoxification and excretion of hydrophobic and xenobiotic metabolites, in the synthesis of most plasma proteins, and in the synthesis, secretion, and conservation of bile acids that are essential for the intestinal absorption of fats and lipids, including fat-soluble vitamins. So, the presence of liver disease is often recognized based on elevated serum activities of liver-derived enzymes such as AST, ALT, LDH and GGT (Hornbuckle and Tennant, 2008). In the present work, the broiler chickens did not present any type of pathology or systemic effect that could be evidenced in the serum. Administration with probiotics in the present trial demonstrated their safety. Pizzolitto et al. (2013) agreed with our results demonstrating that animals fed with a diet containing 1,2 mg AFB1/kg improved biochemical parameters by the application of S. cerevisiae CECT 1891 (5 × 109 cells/L); although they added the yeast in the drinking water.
The liver is the main organ of detoxification and the main target organ of AFB1 (Ortatatli et al., 2005; Mutlu et al., 2010). In this study, the livers of chickens were examined for pathological changes. From the macroscopic examination, a beneficial effect of the use of S. cerevisiae RC016 mixed with L. rhamnosus RC007 occurred because a slightly darker brown colour was seen (Fig. 1, T3 and T4), which suggests a protective effect against aflatoxicosis. Pizzolitto et al. (2012) demonstrated that S. cerevisiae CECT 1891 added to a diet (1010 cells/kg) or to drinking water (109 cells/L) had a beneficial protective effect on the liver histopathological changes of broiler chickens fed AFB1 contaminated diets. It is important to highlight that in the present work, animals received an AFB1 naturally contaminated diet (22.1 ng/kg) that exceeded the recommendation (20 ng/kg) (Alonso et al., 2010) and the slight hydropic degeneration present in control and MA treatments were probably due to this mycotoxin diet contamination. Histopathology of chicken livers with AFB1 showed bile duct proliferation and hepatocellular degeneration as a typical pattern of aflatoxicosis. The absence of microvacuolar fat degeneration suggested the protective effect of the MA in hepatic aflatoxicosis.
Upon ingestion of contaminated feed, the gastrointestinal tract (GIT) is particularly affected by mycotoxin. Generally, the intestinal barrier in the GIT functions as a filter against harmful mycotoxins. However, some mycotoxins have been found to exert detrimental effects on the GIT. For example, mycotoxins can alter normal intestinal functions such as barrier function and nutrient absorption. Some mycotoxins also affect the histomorphology of the intestine (Liew and Mohd-Redzwan, 2018). Akinrinmade et al. (2016) also demonstrated intestinal lesions induced by AFB1 but in rats. In the treatment with AFB1, the infiltration of leukocytes and lymphocytes was observed in the lamina of the intestinal mucosa. In the duodenum and ileum, exposure to AFB1 caused intestinal lesions such as the development of the subepithelial space and degeneration of the villi. Adverse effects in the intestine from exposure to AFB1 include disruption of the intestinal barrier, cell proliferation, cell apoptosis, and the immune system. In the present work, the Brünner glands constitute mucin secreting glandular acini located in the deep and submucosal mucosa layer of the duodenum. They proliferated and probably secreted mucus, pepsinogen, and urogastrone in response to acid stimulation as a response to the presence of AFB1. As in this work, Del Carmen et al. (2013) found a significant decrease in intestinal damage but using a treatment with recombinant lactic acid bacteria in a murine model of colitis.
CONCLUSION
The addition of Saccharomyces cerevisiae RC016 and Lactobacillus rhamnosus RC007 in a MA in broiler chicken’s diets generated innovative beneficial effects. On the one hand, it provided protection against mycotoxin contamination since it maintains adequate levels of the productive parameters avoiding the deleterious effect of AFB1, but also exerting a beneficial influence as probiotic, contributing not only to increasing the productive parameters but also exerting a positive effect on the health, evidenced through an improvement in biochemical and histopathological parameters in broiler chickens. This is of great importance at the economic level for the avian production system.
Acknowledgments
The authors thank SECyT-UNRC, PIP-CONICET GI 11220120100156 and PICT 3089/18 for their funding.
REFERENCES
Abd El-Moneim, A. E. M. E.; El-Wardany, I.; Abu-Taleb, A. M.; Wakwak, M. M.; Ebeid, T. A.; Saleh, A. A. 2020 Assessment of in ovo administration of Bifidobacterium bifidum and Bifidobacterium longum on performance, ileal histomorphometry, blood hematological, and biochemical parameters of broiler chickens. Probiotics and antimicrobial proteins. 12(2):439-450.
Akinrinmade, F.J.; Akinrinde, A.S.; Amid, A. 2016. Changes in serum cytokine levels, hepatic and intestinal morphology in aflatoxin B.-induced injury: modulatory roles of melatonin and flavonoid-rich fractions from Chromolena odorata. Mycotoxin Research. 32:53-60.
Alagawany, M.; El-Hack, M.E.A.; Farag, M.R.; Sachan, S.; Karthik, K.; Dhama, K. 2018. The use of probiotics as eco-friendly alternatives for antibiotics in poultry nutrition. Environmental Science Pollution Research. 25:10611-10618.
Alonso, V.A.; Monge, M.P.; Larriestra, A.; Dalcero, A.M.; Cavaglieri, L.R.; Chiacchiera, S.M. 2010. Naturally occurring aflatoxin M. in raw bulk milk from farm cooling tanks in Argentina. Food Additives Contaminants. 27:373-379.
Arif, M.; Iram, A.; Bhutta, M. A.; Naiel, M. A.; El-Hack, A.; Mohamed, E.; Taha, A. E. 2020. The biodegradation role of Saccharomyces cerevisiae against harmful effects of mycotoxin contaminated diets on broiler chicken’s performance, immunity status, and carcass characteristics. Animals. 10(2): 238.
Armando, M.R.; Dogi, C.A.; Rosa, C.A.; Dalcero, A.M.; Cavaglieri, L.R. 2012. Saccharomyces cerevisiae strains and the reduction of Aspergillus parasiticus growth and aflatoxin B. production at different interacting environmental conditions, in vitro. Food Additives and Contaminants. 29:1443-1449.
Bai, S.P.; Wu, A.M.; Ding, X.M.; Lei, Y.; Bai, J.; Zhang, K.Y.; Chio, J.S. 2013. Effects of probiotic-supplemented diets on growth performance and intestinal immune characteristics of broiler chickens. Poultry Science. 92:663-670.
Blajman, J.; Gaziano, C.; Zbrun, M.V.; Soto, L.; Astesana, D.; Berisvil, A.; Frizzo, L. 2015. In vitro and in vivo screening of native lactic acid bacteria toward their selection as a probiotic in broiler chickens. Research in Veterinary Science. 101:50-56.
Caly, D.L.; D'Inca, R.; Auclair, E.; Drider, D. 2015. Alternatives to antibiotics to prevent necrotic enteritis in broiler chickens: a microbiologist's perspective. Frontiers Microbiology. 6:1336.
Chiocchetti, G.M.; Jadán-Piedra, C.; Monedero, V.; Zúñiga, M.; Vélez, D.; Devesa, V. 2019. Use of lactic acid bacteria and yeasts to reduce exposure to chemical food contaminants and toxicity. Critical Reviews in Food Science and Nutrition. 59:1534-1545.
Dale, N. 1994. National research council nutrient requirements of poultry–ninth revised edition. Journal of Applied Poultry Research. 3(1):101.
Del Carmen, S.; Zurita-Turk, M.; Lima, F.A.; Dos Santos, J.C.; Leclercq, S.Y.; Chatel, J.M.; LeBlanc, J.G. 2013. A novel interleukin-10 DNA mucosal delivery system attenuates intestinal inflammation in a mouse model. European Journal of Inflammation. 11:641-654.
Dogi, C.A.; Fochesato, A.; Armando, R.; Pribull, B.; Souza, M.M.S.; da Silva Coelho, I.; Cavaglieri, L. 2013. Selection of lactic acid bacteria to promote an efficient silage fermentation capable of inhibiting the activity of Aspergillus parasiticus and Fusarium gramineraum and mycotoxin production. Journal Applied Microbiology. 114:1650-1660.
FAO/WHO. 2001 Health and nutritional properties of probiotics in food including powder milk with live lactic acid bacteria. Report of a Joint FAO/WHO Expert Consultation on Evaluation of Health and Nutritional Properties of Probiotics in Food including Powder Milk.
Fochesato, A.S.; Galvagno, M.A.; Cerrutti, P.C.; Gonzalez Pereyra, M.L. 2018. Optimization and Production of Probiotic and Anti-mycotoxin Yeast Biomass Using Bioethanol Industry Waste via Response Surface Methodology. Advanced Biotech Microbiology. 81:555727.
Gadde, U.; Kim, W.H.; Oh, S.T.; Lillehoj, H.S. 2017. Alternatives to antibiotics for maximizing growth performance and feed efficiency in poultry: a review. Animal Health Research Reviews. 18:26-45.
García, G.R.; Payros, D.; Pinton, P.; Dogi, C.A.; Laffitte, J.; Neves, M.; Oswald, I.P. 2017. Intestinal toxicity of deoxynivalenol is limited by Lactobacillus rhamnosus RC007 in pig jejunum explants. Archives of Toxicology. 92:983-993.
Gonzalez Pereyra, M.; Dogi, C.; Torres Lisa, A.; Wittouck, P.; Ortiz, M.; Escobar, F.; Bagnis, G.; Yaciuk, R.; Poloni, L.; Torres, A.; Dalcero, A.; Cavaglieri, L. 2014. Genotoxicity and cytotoxicity evaluation of probiotic Saccharomyces cerevisiae RC016: a 60-day subchronic oral toxicity study in rats. Journal Applied Microbiology. 117(3):824-33.
Hasan, S.; Hossain, M.M.; Alam, J.; Bhuiyan, M.E.R. 2015. Beneficial effects of probiotic on growth performance and hemato-biochemical parameters in broilers during heat stress. International Journal Innovation and Applied Studies. 10:244.
Hornbuckle W.E.; Tennant B.C. 2008. Hepatic Function. Chapter. 15 - Gastrointestinal Function, Clinical Biochemistry of Domestic Animal. 413-457.
Liew, W.P.P.; Mohd-Redzwan, S. 2018. Mycotoxin: its impact on gut health and microbiota. Frontiers in Cellular and Infection Microbiology. 8:60.
Magnoli, A.P.; Monge, M.P.; Miazzo, R.D.; Cavaglieri, L.R.; Magnoli, C.E.; Merkis, C.I.; Cristofolini, A.L.; Dalcero, A.M.; Chiacchiera, S.M. 2011. Effect of low levels of aflatoxin B. on performance, biochemical parameters, and aflatoxin B. in broiler liver tissues in the presence of monensin and sodium bentonite. Poultry Science. 90:48–58.
Magnoli, A.P.; Rodriguez, M.C.; Pereyra, M.G.; Poloni, V.L.; Peralta, M.F.; Nilson, A.J.; Cavaglieri, L.R. 2017. Use of yeast (Pichia kudriavzevii) as a novel feed additive to ameliorate the effects of aflatoxin B. on broiler chicken performance. Mycotoxin Research. 33:273-283.
Mehdi, Y.; Létourneau-Montminy, M.P.; Gaucher, M.L.; Chorfi, Y.; Suresh, G.; Rouissi, T.; Godbout, S. 2018. Use of antibiotics in broiler production: Global impacts and alternatives. Animal Nutrition. 4:170-178.
Mutlu, A.G.; Kursun, O.; Kasİmoglu, A.; Dukel, M. 2010. Determination of aflatoxin M. levels and antibiotic residues in the traditional Turkish desserts and ice creams consumed in Burdur city center. Journal of Animal and Veterinary Advanced. 9:2035-2037.
Ortatatli, M.; Oguz, H.; Hatipoglu, F.; Karaman, M.X. 2005. Evaluation of pathological changes in broilers during chronic aflatoxin (50 and 100 ppb) and clinoptilolite exposure. Research Veterinary Science. 78:61-68.
Park, S.H.; Lee, S.I.; Ricke, S.C. 2016. Microbial populations in naked neck chicken ceca raised on pasture flock fed with commercial yeast cell wall prebiotics via an Illumina MiSeq platform. PLoS One. 11(3): e0151944.
Peng, Q.Y.; Li, J.D.; Li, Z.; Duan, Z.Y.; Wu, Y.P. 2016. Effects of dietary supplementation with oregano essential oil on growth performance, carcass traits and jejunal morphology in broiler chickens. Animal Feed Science and Technology. 214:148-153.
Pizzolitto, R.P.; Salvano, M.A.; Dalcero, A.M. 2012. Analysis of fumonisin B. removal by microorganisms in co-occurrence with aflatoxin B. and the nature of the binding process. International Journal of Food Microbiology. 156:214-221.
Pizzolitto, R.P.; Armando, M.R.; Salvano, M.A.; Dalcero, A.M.; Rosa, C.A. 2013. Evaluation of Saccharomyces cerevisiae as an antiaflatoxicogenic agent in broiler feedstuffs. Poultry Science. 92:1655-1663.
Poloni, V.; Magnoli, A.; Fochesato, A.; Cristofolini, A.; Caverzan, M.; Merkis, C.; Cavaglieri, L. 2020. Saccharomyces cerevisiae RC016-based feed additive reduces liver toxicity, residual aflatoxin B. levels and positively influences intestinal structure in broiler chickens fed on chronic aflatoxin B. levels-contaminated diets. Animal Nutrition. 6:31-38.
Ronquillo, M.G.; Hernandez, J.C.A. 2017. Antibiotic and synthetic growth promoters in animal diets: review of impact and analytical methods. Food Control. 72:255-267.
Śliżewska, K.; Cukrowska, B.; Smulikowska, S.; Cielecka-Kuszyk, J. 2019. The effect of probiotic supplementation on performance and the histopathological changes in liver and kidneys in broiler chickens fed diets with aflatoxin B1. Toxins. 11:112.
Trucksess, M.W.; Stack, M.E.; Nesheim, S.; Albert, R.; Romer, T. 1994. Multifunctional column coupled with liquid chromatography for determination of aflatoxins B., B., G. and G. in corn, almonds. Brazil nuts, peanuts, and pistachio nuts: Collaborative study. Journal AOAC International. 77:1512-1521.
Vuong, C.N.; Chou, W.K.; Hargis, B.M.; Berghman, L.R.; Bielke, L.R. 2016. Role of probiotics on immune function and their relationship to antibiotic growth promoters in poultry, a brief review. International Journal of Probiotics and Prebiotics. 11:1-7.
Fochesato, A.S.; Cuello, D.; Poloni, V.; Galvagno, M.A.; Dogi, C.A.; Cavaglieri, L.R. 2019. Aflatoxin B1 adsorption/desorption dynamics in the presence of Lactobacillus rhamnosus RC007 in a gastrointestinal tract‐simulated model. Journal Applied Microbiology. 126:223-229.
Author notes
lcavaglieri@exa.unrc.edu.ar

