Abstract: Species of the genus Senecio have been traditionally employed with medical purposes and it has been demonstrated that some of them have anti-proliferative activity on cancer-derived cell lines. In South America S. madagascariensis is one of the most important representatives of the genus. Almost all species contain pyrrolizidine alkaloids (PA), substances that can lead to hepatic damage. Thus, the aim of this work was to determine the cytotoxic activity of a low PAs extract of S. madagascariensis on a cancer-derived cell line. A chloroform extract from plant material was produced and it was demonstrated to have a PAs concentration lower than 200 µg/g (w/w). Two stock solutions were made with DMSO and acetone as solvents and were diluted in culture medium (DMEM). Cells from urinary bladder transitional cell carcinoma T24 were cultured in 96 well plates (104 cells/well) and incubated for 24 hours with dilutions of the extracts at the following concentrations, 0.5 mg/mL, 0.25 mg/mL, 0.125 mg/mL and 0.06 mg/mL. Cell viability was evaluated by the MTT colorimetric technique. Both solutions of S. madagascariensis extracts (acetone and DMSO) showed cytotoxic activity. In both cases, the degree of cytotoxicity was dependent on the concentration of the solution.
Keywords: anti-proliferative activity, cytotoxic activity, phytomedicine, senecio madagascariensis, medicinal plants.
Resumen: Las especies del género Senecio han sido empleadas tradicionalmente con fines medicinales y se ha demostrado que algunas de ellas poseen actividad antiproliferativa en células cancerosas. En América del Sur, S. madagascariensis es uno de los representantes más importantes del género. La mayoría de las especies contienen alcaloides pirrolizidínicos (AP), sustancias que pueden provocar daño hepático. Por lo tanto, el objetivo de este trabajo fue determinar la actividad citotóxica de un extracto bajo en AP de S. madagascariensis en una línea celular cancerosa. Se desarrolló un extracto clorofórmico a partir del material vegetal con una concentración de AP menor a 200 µg/g (w/w). Dos soluciones stock fueron realizadas utilizando DMSO y acetona como solventes y fueron diluidas en medio de cultivo (DMEM). Fueron cultivadas células de carcinoma de células transicionales de vejiga urinaria T24 en placas de 96 pocillos (104 células por pocillo) e incubadas por 24 horas con diluciones de los extractos a las siguientes concentraciones: 0,5 mg/mL, 0,25 mg/mL, 0,125 mg/mL y 0,06 mg/mL. La viabilidad celular fue evaluada mediante la técnica colorimétrica de MTT. Ambas soluciones de S. madagascariensis demostraron actividad citotóxica. En ambos casos el grado de citotoxicidad fue dependiente de la concentración de la solución.
Palabras clave: actividad antiproliferativa, actividad citotóxica, fitomedicina, Senecio madagascariensis, plantas medicinales.
Resumo: As especies do gênero Senecio têm sido tradicionalmente utilizadas para fins medicinais e algumas de las mostraram ter atividade antiproliferativa em células cancerosas. Em América do Sul, S. madagascariensis é um dos representantes mais importantes do gênero. A maioria das espécies contém alcalóides pirrolizidínicos (PAs), substâncias que podem causar dano ao fígado. Portanto, o objetivo deste trabalho foi determinar a atividade citotóxica de um extrato de S. madagascariensis com baixo teor de PA em uma linhagem de células cancerosas. Um extrato clorofórmico foi realizado a partir do material vegetal com uma concentração de PA inferior a 200 µg/g (w/w). Dois soluções estoque foram feitas usando DMSO e acetona como solventes e foram diluídas em meio de cultura (DMEM). Células de carcinoma de células transicionais da bexiga urinária T24 foram cultivadas em placas de 96 poços (104 células por poço) e incubadas por 24 horas com diluições dos extratos nas seguintes concentrações 0,5 mg / mL, 0,25 mg / mL, 0,125 mg / mL e 0,06 mg / mL. A viabilidade celular foi avaliada pela técnica colorimétrica MTT. Ambas soluções de S. madagascariensis demonstraram atividade citotóxica. Em ambos casos, o grau de citotoxicidade foi dependente da concentração da solução.
Palavras-chave: atividade antiproliferativa, atividade citotóxica, fitomedicina, senecio madagascariensis, plantas medicinais.
Animal production and pastures
Cytotoxic effect of Senecio madagascariensis (Asteraceae) extracts on cancer derived cell lines
Efecto citotóxico de extractos de Senecio madagascariensis (Asteraceae) en lineas celulares derivadas de cáncer
Efeito citotóxico de extratos de Senecio madagascariensis (Asteraceae) em linhagens celulares derivadas de câncer
Received: 09 October 2020
Accepted: 20 October 2021
Published: 25 November 2021
Corresponding author: cgarciaysantos@fvet.edu.uy

Plants as source of medicinal products have been used since ancient times. Fossil records date this type of medicine at least 60.000 years ago(1). Currently, this traditional medicine is being reconsidered worldwide and significant research activity on the therapeutic properties of plant species is being carried out(2)(3)(4). Plants of the family Asteraceae have an enormous importance in popular medicine(5)(6)(7). This family, also known as Compositae, is one of the largest families of plants and is constituted by 23,600 species(8). The specimens can be found widespread all over the world(9). Within Asteraceae family, Senecio is the largest genus(10). Species of Senecio have been traditionally employed as anti-inflammatory, anti-emetic and vasodilator agents(11)(12). More recently, it has been demonstrated that some species of this genus, like S. vulgaris, S. inaequidens, S. stabianus and S. tenuifolius, have anti-proliferative activity in cancer derived cells(10)(13)(14). In South America, particularly in Brazil, Uruguay and Paraguay, more than 90 species of Senecio have been reported, being S. brasiliensis, S. madagascariensis and S. selloi the most important(15)(16)(17). As far as we know, no reports of anti-proliferative activity on cancer derived cell lines of those species have been published. Almost all of the Senecio species contain pyrrolizidine alkaloids (PAs) as the main metabolites(18), but chalcones and flavonoids have also been reported(19). PA become toxic compounds when bioactivated by mixed function oxidases to toxic dehydropyrrolizidine alkaloids (pyrroles). Most of this activation is done in the liver, leading to hepatic damage(20). Therefore, the medical use of Senecio extracts with PAs would not be recommended, but, although pyrrolizidines are cytotoxic and antimitotic, the anti-cancer properties of Senecio species could be due the action of multiple compounds and not PAs exclusively.
Consequently, the aim of this work was to determine the cytotoxic activity of an extract of Senecio madagascariensis with undetectable amounts of PAs on a cancer-derived cell line.
Plant material was sampled from the rural area of Montevideo, Uruguay (latitude: 34°45′30´´S; longitude: 56°18′08´´W). The above ground parts of the plants were collected during winter (July and August) of 2016. Plant identification was confirmed by the Botanic Laboratory of the Agronomic School of the University of the Republic, and deposited in their herbarium (MVFA).
The plant material was air-dried in the dark and grinded to a thick powder. Samples (10 g) were macerated with acidified CHCl3 (100 mL) for 7 days in order to obtain the extract. After this, extract was evaporated in vacuum, lyophilized and kept at −20°C until its use. In order to be used this lyophilized extract had to be solved in different solvents, as explained below.
TLC scanning determinations were performed on a Shimadzu (Tokyo, Japan) Model 9300 TLC scanner at 505 nm for Dragendorff reagent, using the reflection system in the zig-zag mode with an 8 mm swing width.
The quantification of the PAs was carried out by a modification of the TLC-Scanning method previously developed in our laboratory(21). Senecionine was used as standard. In brief, aliquots (20µL) of the extract solutions in CHCl3 (10 mg/mL) were disposed by duplicate on the TLC plate and the same volume of the working standard solution at increasing concentrations (0.05 – 2 mg/mL) was added. The plate was developed to 10 cm in a 17.5 cm × 11.0 cm × 6.2 cm chamber (saturation time 30 min), using CHCl3/MeOH 90:10 as the mobile phase, sprayed with Dragendorff reagent, air-dried and submitted to scanning.
Two stock solutions of the extract were prepared using two different solvents, acetone and dimethyl sulfoxide (DMSO), both at the concentration of 100 mg/mL. The objective of this was to determine which solvent worked better to dissolve the lyophilized extracts. Solutions were then diluted with culture medium (DMEM) yielding final extracts concentrations with DMSO or acetone concentration of 0.5%.
A cell line of urinary bladder transitional cell carcinoma T24 (ATCC No.: HTB-4) was cultured in Dulbecco's modified Eagle's medium (DMEM) low glucose, supplemented with 10% fetal bovine serum (FBS; Capricorn scientific, South America) and 1% antibiotic (penicillin 107 Unit/L, streptomycin 10000 mg/L; Capricorn scientific, Germany) in tissue culture flasks (Greiner, Germany). The cultures were incubated at 37°C with 5% CO2 and the medium was changed every 48 hours. When 80% to 90% confluence was reached, the cultures were treated with trypsin-ethylenediaminetetraacetic acid (EDTA) 0.004% (SAFC, USA) and subcultured(22).
Cells were washed with phosphate buffer saline solution (PBS), collected by trypsinization, plated in 96 well plates (104 cells/well) and incubated under 5% CO2 at 37ºC for 24 hours. After that, the cells were treated with the DMSO and acetonic Senecio extract dilutions at the following concentrations, 0.5 mg/mL, 0.25 mg/mL, 0.125 mg/mL and 0.06 mg/mL.
Figure 1 schematizes the process from the plant extraction to the cell treatment. Control cells were incubated in culture medium only. Additionally, in order to evaluate if the solvents affected the viability of the cells by themselves, a control group of cells were incubated with acetone 0.5% (CA) and DMSO 0.5% (CD).
All concentrations of plants extract and controls were evaluated in quadruplicated on the same cell batch of 96 wells. The cells were incubated with the extracts for 24 hours. Cell viability was evaluated by the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay(23).
This assay is based on cleavage of the yellow tetrazolium salt, MTT, which forms water-insoluble, dark blue formazan crystals. This cleavage only takes place in alive cells by the enzyme succinate-dehydrogenase located within the mitochondria. After 24 hours of incubation, medium was removed and 15 μL of MTT solution (5 mg/mL) were added on each well (0.1 mg/mL final concentration). The plates were incubated for 4 hours under 5% CO2 at 37ºC. MTT reagent was removed, and the formazan crystals were dissolved in 100 μL of DMSO. The absorbance was determined in a microplate reader (Varioskan Flash, Thermo Scientific), using a reference wavelength of 630 nm and a test wavelength of 570 nm. Results are presented as percentage of MTT reduction, assuming that the control absorbance was 100%.
The percentage of growth inhibition was calculated using the following formula:
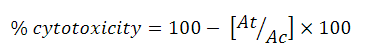
Where At = absorbance value of test compound, and Ac = Absorbance value of control. The effects of extracts were expressed by IC50 values (the drug concentration needed to reduce the absorbance of the treated cells by 50% in comparison with untreated cells).
All results are expressed as mean percentage of cytotoxicity relative to the control ± SEM. All measurements were replicated three times at different days. The percentage of cell inhibition was transformed by the arcsine square root transformation and analyzed by one-way ANOVA, and p < 0.05 was considered statistically significant. Graph Pad Prism 9.0 (San Diego, USA) was used to perform the statistical analysis.
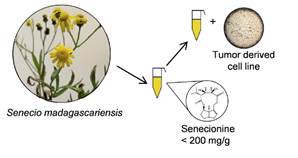
Schematic representation of the process from the plant recollection, the obtention of the extracts, the quantifications of PAs and the application of these extracts on cancer derived cell-lines culture.
Available data: The entire data set that supports the results of this study was published in the article itself.
The dried extracts of S. madagascariensis leaves were analyzed by TLC-scanning and the concentration of PAs was undetectable. Since this method has a limit of quantification of 0.2% (signal-to-noise ratio 2), the obtained extracts had a PAs concentration lower than 200µg/g (w/w) expressed as senecionine. The extract was diluted in acetone and DMSO at 0.5 mg/mL in the most concentrated solutions, being the effective concentration of PAs lower than 1 µg/mL.
In the control groups, DMSO or acetone at the concentration of 0.5 mg/mL did not affect cell viability when compared with the control cells incubated in culture medium only. Being the mean ± standard deviation absorbance of the control group with culture medium, CA and CD 1.20 ± 0.02, 1.18 ± 0.08 and 1.09 ± 0.2, respectively. The results of a one-way ANOVA were F(2,7) = 0.5442, p = 0.6030. Consequently, we ruled out the possibility that the solvents could affect the cells viability. Cells treated with acetonic solutions of the extract were then compared to the CA group of cells, and the ones treated with DMSO solutions were compared to CD group.
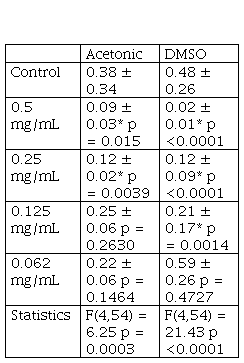
Both solutions of S. madagascariensis extracts (acetonic and DMSO) showed cytotoxic activity. In both cases, the degree of cytotoxicity was dependent of the concentration of the solution. The absorbance values of each extract dilution and the statistical analysis results are shown in Table 1.
Regarding the acetonic solution, 0.5 mg/mL and 0.25 mg/mL had significant cytotoxic activity with respect to the control, while the effects of the additional dilutions were not statistically significant (Figure 2A). The effect of DMSO solution of the Senecio extract is shown in Figure 2B, the concentrations with significant cytotoxicity compared to CD were 0.5 mg/mL, 0.25 mg/mL and 0.125 mg/mL. The more diluted solutions did not have an effect on the cell viability.
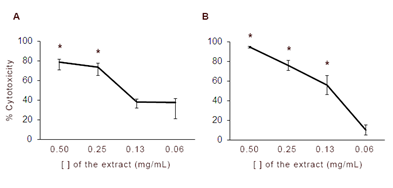
Cytotoxic activity of acetonic (A) and DMSO (B) dilutions of a chloroform S. madagascariensis extract. Percentage of cytotoxicity curve. Percentage of cytotoxicity (100−(absorbance of test compound/absorbance of control) ×100) plotted against the concentration of extract. The asterisk indicates significant differences with respect to the control group (p < 0.05).
We demonstrated that chloroform extracts of S. madagascariensis diluted in both acetone and DMSO have cytotoxic properties on cancer-derived cell lines. Screening models of cytotoxic activity provide valuable preliminary data to select plant extracts with potential antineoplastic properties(24). We evaluated two different solvents for the chloroform extract, acetone and DMSO; and DMSO extract seemed to have higher % of cytotoxicity at the same extract concentrations, none of the solvents showed intrinsic cytotoxic activity at the employed concentration. Our result is in accordance with other reports of antineoplastic cytotoxic and antineoplastic activity of Senecio species not so common in South America(10)(13)(14)(25). To the best of the authors' knowledge, this is the first report of cytotoxic activity of an extract of S. madagascariensis in cancer derived cell lines. S. madagascariensis is known by its hepatotoxic activity. In fact, poisoning by different species of Senecio is one of the leading causes of death in cattle in extensive production systems in Uruguay and Brazil(26)(27). It has been proved that the hepatotoxicity of Senecio is due to the presence of PAs(20). However, in this study we aimed to evaluate the cytotoxic effect of a S. madagascariensis extract that could potentially be studied in the future in preclinical and clinical studies for cancer treatment, therefore, we used an extract with an undetectable PAs concentration, i.e., without it’s known toxic compound.
This study intends to be a preliminary report of the cytotoxic activity of a chloroform extract of the plant and, has the limitation of not having studied the composition of the extract and its potential toxicity in normal cells. Further basic studies are needed before planning preclinical trials. Other studies have found that sesquiterpenes in Senecio are responsible for their cytotoxic effect in cancer derived cell lines(25)(28)(29). Interestingly, sesquiterpenes have shown telomerase inhibition properties(25)(28). This enzyme is a reverse transcriptase that synthesizes telomeric repeats. It is found in germ line cells and stem cells, and cancer-derived cells, but is very rare in adult cells in most somatic tissues.
Because 80% of tumors express telomerase, it has been suggested that the inhibition of telomerase can be a target of antineoplastic drugs in order to selectively affect the cancer-derived cells without affecting normal cells(30). Sesquiterpenes derivates have been found in S. madagascariensis, but its cytotoxic properties have not been evaluated yet(31). Other studies have found that jacaranone phytoquinoide is the responsible of the cytotoxic properties of Senecio species(24)(32). Another limitation of the study is that the cytotoxicity of the extract was only evaluated in one specific cancer derived cell line, cells derived from urinary bladder transitional cell carcinoma. Therefore, we cannot rule out the possibility that this effect is specific for this cell line. However, other studies with Senecio species have found cytotoxic activity in other cell lines. For example, Tundis and others(10) demonstrated the cytotoxic activity of S. stabianus against renal adenocarcinoma, hormone-dependent prostate carcinoma, amelanotic melanoma and human breast adenocarcinoma cell lines, and no selectivity for a specific cell line was found. Additionally, S. oldhamianus extracts were effective against human hepatoma cells and human oophoroma cells, again without showing any selectivity(25). Consequently, while further studies with different cell lines are required, according to the literature, it seems that Senecio extracts are active against different populations of cancer derived cell lines.
Finally, it is worth to consider that S. madagascariensis is a very invasive plant, exceedingly difficult to control by conventional control methods; it usually requires physical removal of its root system(17)(33). Further studies are required to demonstrate its potential anticarcinogenic activity, but if demonstrated, it will be a plant with medical properties very easy to find in South America, and the waste produced by mechanical removal of the plants could be exploited.
This study intended to be a preliminary screening of anti-proliferative activity of S. madagascariensis on a cancer-derived cell line, and therefore it has the limitations of analyzing the effect on only one kind of cell line, urinary bladder transitional cell carcinoma. Thus, these extracts should be tested in other various cell types. Therefore, we conclude that at a preliminary stage, PAs free S. madagascariensis extracts possess cytotoxic activity against urinary bladder transitional cell carcinoma.
Author contribution
statement: AM conceived and designed the
analysis, performed the experiments and collected the data, did the analysis,
and wrote the manuscript. KY designed the analysis, performed the experiments,
reviewed and edited the manuscript. AI performed the experiments and collected
the data, reviewed and edited the manuscript. CE helped in designing the
experiments and collecting data, and supervised, reviewed and edited the
manuscript. SR performed experiments and collected data. AG performed
experiments and collected data, reviewed and edited the manuscript. CG
conceived and designed the analysis, supervised, acquired the finances,
reviewed and edited the manuscript.
Editor: The following
editor approved this article.
Laura Astigarraga (https://orcid.org/0000-0002-7696-7539)
Universidad de la República, Facultad de Agronomía, Montevideo, Uruguay.
http://agrocienciauruguay.uy/ojs/index.php/agrociencia/article/view/425/1059 (pdf)
The authors would like to thank Mr. Daniel Pérez for his selfless contribution to society through promoting scientific research aimed at solving community health problems. We also appreciate the collaboration of Dr. Mauricio Bonifacino by the identification of the plant material and Dr. Paola Scavone by sharing with us the tumoral cell line.
cgarciaysantos@fvet.edu.uy

Schematic representation of the process from the plant recollection, the obtention of the extracts, the quantifications of PAs and the application of these extracts on cancer derived cell-lines culture.


Cytotoxic activity of acetonic (A) and DMSO (B) dilutions of a chloroform S. madagascariensis extract. Percentage of cytotoxicity curve. Percentage of cytotoxicity (100−(absorbance of test compound/absorbance of control) ×100) plotted against the concentration of extract. The asterisk indicates significant differences with respect to the control group (p < 0.05).

