



Plant protection
Floral food resources for Apis mellifera (Hymenoptera: Apidae) in a mountain forest area in Uruguay
Flora alimenticia disponible para Apis mellifera (Himenoptera: Apidae) en un área de bosque serrano en Uruguay
Flora alimentar disponível para Apis mellifera (Himenoptera: Apidae) em uma área de floresta de altitude no Uruguai
Agrociencia Uruguay
Universidad de la República, Uruguay
ISSN-e: 2730-5066
Periodicity: Bianual
vol. 25, no. 2, e426, 2021
Received: 13 October 2020
Accepted: 11 March 2021
Published: 14 June 2021
Corresponding author: heihoracio@gmail.com

Abstract: Knowing the blooms is useful to predict the behavior of Apis mellifera (honeybee) colonies. The objective was to determine the floristic characteristics of a mountain forest in Uruguay from the beekeeping point of view. We made eight visits in every three sites surrounding the apiary, at 10-20 m, 500 m and 1500 m distance. The first visit determined the species, families, origin (native or exotic), and frequency of each taxon. In subsequent visits, we censed which ones remained in bloom (presence or absence). We assigned each species a value of one to five for abundance (A) and the duration of flowering (P), during two years. Using the Shannon Index the floristic diversity was established. A generalized linear model was employed to analyze flowering through the response variables: presence of flowering in winter, spring, summer and autumn, as well as flowering duration. We calculated a Shannon Index of 3.6, indicating high biodiversity. Then we correlated the findings of the survey and literature reports on the occurrence of each species in melissopalynological studies in mature and immature honey. The expression “Convertible Flora” was coined to identify the floral resources whose pollen is stored when no flower is available. These were Lithraea brasiliensis, Baccharis articulata, Baccharis trimera, Blepharocalyx salicifolius, Eugenia uniflora, Colletia Paradoxa, Oxalis sp., Scutia buxifolia, Jodina rhombifolia, Aloysia gratissima for the spring and autumn seasons (P <0.05). The expression “Support Flora” (P <0.05) is used to classify those resources that are found in immature honey and are almost absent in mature honey, and are used as nutrients during the colony expansion: Schinus engleri, Maytenus ilicifolia, Eryngium pandanifolium, Baccharis punctulata, Abutilon pauciflorum, Daphnopsis racemosa, Allophylus edulis, and Celtis tala. This flora classification allows planning the expected developments and yields of honey from a region to establish technological useful and efficient packages of honey harvests.
Keywords: Apis mellifera, honeybee calendars, support flora, convertible flora.
Resumen: El conocimiento de las floraciones es útil para predecir el comportamiento de las colonias de Apis mellifera (abeja de la miel). El objetivo fue determinar las características florísticas desde el punto de vista apícola de un bosque serrano en Uruguay. Realizamos ocho visitas en tres sitios aledaños a la colmena: a 10-20 m, 500 m y 1500 m de distancia. En la primera visita determinamos especies, familias, origen (nativo o exótico) y frecuencia de cada taxón. En visitas posteriores, censamos cuáles estaban florecidas (presencia o ausencia). Asignamos a cada especie un valor de uno a cinco para la abundancia (A) y la duración de la floración (P), durante dos años. Utilizamos el índice de Shannon para medir la diversidad florística. Se utilizó un modelo lineal generalizado para analizar la floración a través de las variables respuesta: presencia de floración en invierno, primavera, verano y otoño, así como duración de la floración. Calculamos un índice de Shannon de 3,6, lo que indica una alta biodiversidad. Luego correlacionamos los hallazgos de la prospección con datos de la literatura sobre la ocurrencia de cada especie en estudios melisopalinológicos en mieles maduras e inmaduras. Por último, definimos el término flora convertible para identificar los recursos florales cuyo polen se almacena cuando no hay ninguna flora disponible. Estas fueron Lithraea brasiliensis, Baccharis articulata, Baccharis trimera, Blepharocalyx salicifolius, Eugenia uniflora, Colletia Paradoxa, Oxalis sp., Scutia buxifolia, Jodina rhombifolia, Aloysia gratissima, para las temporadas de primavera y otoño (P<0.05). El término flora de soporte (P<0.05) lo definimos para clasificar aquellos recursos que se encuentran en la miel inmadura y están casi ausentes en la miel madura, y se utilizan como nutrientes durante la expansión de la colonia: Schinus engleri, Maytenus ilicifolia, Eryngium pandanifolium, Baccharis punctulata, Abutilon pauciflorum, Daphnopsis racemosa, Allophylus edulis y Celtis tala. Esta clasificación de la flora permite planificar los desarrollos y rendimientos de la miel esperados de una región, y establecer paquetes tecnológicos útiles y eficientes de cosechas de miel.
Palabras clave: Apis mellifera, calendarios apícolas, flora de soporte, flora convertible.
Resumo: O conhecimento das flores é útil para prever o comportamento de colônias de Apis mellifera (abelha do mel). O objetivo foi determinar as características florísticas de uma floresta montanhosa no Uruguai do ponto de vista apícola. Fizemos oito visitas em três locais no entorno do apiário: a 10-20m, 500m e 1500m de distância dele. A primeira visita determinou as espécies, famílias, origem (nativa ou exótica) e frequência de cada táxon. Nas visitas subsequentes, censuramos quais permaneceram em flor (presença ou ausência). Atribuímos a cada espécie um valor de um a cinco para abundância (A) e duração da floração (P), durante dois anos. Usando o Índice de Shannon, a diversidade florística foi estabelecida. Um modelo linear generalizado foi empregado para analisar a floração através das variáveis resposta: presença de floração no inverno, primavera, verão e outono, bem como a duração da floração. Calculamos um Índice de Shannon de 3,6, indicando alta biodiversidade. Após correlacionar os achados do levantamento e relatos da literatura sobre a ocorrência de cada espécie em estudos melisopalinológicos em mel maduro e imaturo. O termo “Flora Conversível” foi cunhado para identificar os recursos florais cujo pólen é armazenado quando nenhuma flor está disponível. Estes foram Lithraea brasiliensis, Baccharis articulata, Baccharis trimera, Blepharocalyx salicifolius, Eugenia uniflora, Colletia Paradoxa, Oxalis sp., Scutia buxifolia, Jodina rhombifolia, Aloysia gratissima para as estações de primavera e outono (P <0,05). O termo “Flora de Apoio” (P <0,05) é usado para classificar os recursos encontrados no mel imaturo e quase ausentes no mel maduro e são usados como nutrientes durante a expansão da colônia: Schinus engleri, Maytenus ilicifolia, Eryngium pandanifolium, Baccharis punctulata, Abutilon pauciflorum, Daphnopsis racemosa, Allophylus edulis and Celtis tala. Esta classificação da flora permite planejar os desenvolvimentos e rendimentos esperados do mel de uma região para estabelecer embalagens tecnológicas úteis e eficientes de colheita de mel.
Palavras-chave: Apis melífera, calendários de abelhas, flora de suporte, flora conversível.
1. Introduction
Apis mellifera (Honeybees) are eusocial insects(1)(2). They collect carbohydrates, proteins, minerals, and vitamins from flowers, which are essential for their biological development(1)(3)(4). Their eating habits are more selective than other eusocial insects, such as Bombus gerstaekeri(5). Although these are both insects that always visit the same type of flowers (Flower constancy)(5)(6), the honeybees first analyze the nutritional quality of them and then trigger the massive collection of the colony in the selected source. While other insects visit the same flowers only because of the shape, for example, tubular corolla, regardless of the food quality offered(5).
According to Simcock and others(7), honeybees show sensitivity to amino acids in nectar, and preferentially drink those solutions containing amino acids over pure sucrose solutions, presumably differentiating between the two rewards through pre-digestive mechanisms and the nutritional state of the colony(8). This fact restricts visits to a type of flower once it qualifies as the best (nectar or pollen)(5). Then, the honeybee organizes its collection with the rest of the colony´s collectors and begin the selected food search(8)(9). Finally, they take it to a particular place, the nest, whether to consume it directly, store or age it, to transform it into honey for further consumption. They collect the supply of floral resources throughout the seasons(10). This constitutes the beehive’s food storage for the period when there are no flowers that can also be used for colony growth(11). In such cases, it does not remain for a long time in the colony. Honeybees choose four or five species to feed themselves and store as a food reserve in the hive(12).
Forage's flight distance is one of the highest costs in terms of time and energy that a worker honeybee must weigh against the food's gain(13). They have evolved exceptional sensitivity to measuring relative energetic rewards and to discovering new forage sources in the landscape, so a forager will only recruit another honeybee inside hives for the best forage sites known at any given time(14). Therefore, each communication between explorer honeybees and recruited honeybees inside hives has already passed a cost-benefit analysis(15). This means that explorer bees will define an average feeding area as close to the hive as possible, seeking the best efficiency in food collection. Then, they communicate it to the other forager bees directing the foraging efforts within an average radius of influence. The mean feeding radius of bees ranges from several hundred meters to 1500 m, (up to 5500 m) depending on the spatial and temporal context. According to Beekman and Ratniek(13), the flight distance from the hive to the foraging patch affects feeding costs in terms of time, energy expenditure, and grazing mortality. When the landscape is made up of small patches with high diversity over time, foragers can quickly gather food in all types of patches without travelling long distances, and colonies benefit from the diversity of vegetation near the nest(16).
Steffan-Dewenter and others(17) studied the effects of plant's context of landscapes on bees in experimental plots with a varying radius of 250, 500, 750, 1000, 1500, 2000, 2500 and 3000 m, which were selected based on known flight and foraging distances for solitary bees and honeybees. They determined that the floral diversity positively influences bees' permanence in a region and has more temporal stability. The honeybees are sustainable in habitats with a diversity of species(18). The more diverse(19) and rich flora a zone has, the more influential is on the bees population dynamic(20)(21). The knowledge and characterization of the environment's blooms allow us to predict the behavior, development, and maintenance of bee colonies(14).
The most common methodology to determine the botanical origin of nectar and the contribution of nectar-pollen resources used by hives is the melissopalynological study(22). The analysis of the types of pollen present in honey makes it possible to know the flowers selected by bees and contributes to decision making protocols that improve beekeeping. The result obtained through these analyses in mature honey indicates the floral diversity, forest vegetation and composition of plant species that honeybees visit to get and extract nectar to mature and store later in the hive. On the other hand, it is possible to detect the plants used by honeybees analyzing nectar in the hive (immature honey), which will be consumed during the colony's growth period(11) .
The present study aimed to determine the floristic characteristics of a mountain forest region in Lavalleja, Uruguay, throughout the seasons, during two years. Specifically, we determined the food resource availability for honeybees. This study's results show the potential of this type of regions for apicultural activities in the future.
2. Materials and methods
2.1 Study area and sampling
The work began in March 2014, on a property located between route N 8 and N 81, Uruguay (34º 54' 01, 34" S, 55° 31' 19, 18" W). Within the region, we selected a mountain forest with groups of various native woody plant species; with a strongly hilly profile with a slope that oscillates between 12 and 25% and poorly developed soil, with a neutral to moderately alkaline pH. The climate is warm subtropical serrano(23) with average temperatures of 25°C in summer and 13°C in winter, with an average annual rainfall of 800-1000 mm. Precipitation is the lowest in December, averaging 85 mm, and 124 mm is the average fall in April, the month with the highest rainfall of the year. From the phytogeography point of view, the vegetation belongs to the mountain forest of Lavalleja Department. Mountain forests occupy a vast area in Uruguay and have a high floral diversity(24). Some characteristic taxa are Heterothalamus alienus and Baccharis sp. Along with shrubs such as Dolichandra unguis-cati and Daphnopsis racemosa. The trees are mainly by Lithraea brasiliensis, Schinus sp. and Myrtaceae. Lithraea brasiliensis is one of the most abundant species in these forests. Myrtaceae represents the species Blepharocalyx salicifolius and Myrrhinium loranthoides, Eugenia uruguayensis, Eugenia uniflora and Myrceugenia glaucescens(24). Due to its topographic and edaphic characteristics, it is an area not suitable for agriculture, but in general, it supports the development of low forestry activity, especially of Eucalyptus species, accompanied by subsistence livestock. In this area, beekeeping is carried out commercially with a prevalence of diverse and stable vegetation(25), without agriculture that might support an ecological niche for honeybees.
We surveyed floral species found within the flight radius of a bee under normal conditions, estimated as 1.5 km, since this work aimed to evaluate plant species from a beekeeping point of view(13). We marked three randomly chosen and zigzag transects sites over the area of influence in one apiary with 20 hives. We located transect one at a 10-20 meters, site two at 500 m, and three at 1.500 m. Each site was 3 m wide by 20 m long (180 m²) (0.018 ha). The sampling area represented 0.01% of the foraging area. We sampled in eight opportunities during the morning (8 am to 1 pm), over two years, each during a different middle season (winter, spring, summer, and autumn) to measure plant attributes floristic composition: taxonomy, frequency, abundance and flowering time.
2.2 Plant attributes
Firstly, to check the floral diversity of the region, we characterized in the first visit each taxon by botanic family and botanic species and we determined from those not initially identified at the site through comparison with arboretum samples. We estimated the similarity between species in terms of floristic composition by the species accumulation curve, with the Shannon Index. This index evaluates floral heterogeneity based on two factors: the number of floral species present and their relative abundance(26). We studied the richness of the species by the species accumulation method(27). The heterogeneity of plants given by the floristic composition registered identifies the site in a specific category of vegetation. It demonstrates the richness of plant species of a particular type of vegetation calculated as the sum of all the different species recorded in each transect or plots(28)(29). The species' diversity had two main components: number of botanic species and fairness (number of individuals of each species) frequency, and abundance. In subsequent visits over two years, we assessed the presence and duration of flowering (yes or not). We rated the species surveyed in relation to the abundance (A) with scale = 1 (Rare = one individual); 2 (Minor importance = two-five individuals); 3 (Secondary = six-eight individuals); 4 (Abundant = nine-12 individuals); 5 (Very Abundant = more than 12 individuals).
We studied the time they remained in bloom through the seasons with the following scale (P) = 1 (Less than one season blooming); 2 (One flowering season); 3 (Two flowering season); 4 (Three flowering seasons); 5 (All year flowering) (Table 1). Also, we determined and applied the term Native (N) to the natural plants of Uruguay not introduced or naturalized. On the contrary, Exotic (E) is the term for an introduced plant.
Then, we searched for literature that reported honeybee pollen in samples of both mature and immature honeys in order to characterize the plants found in the ecoregion as suppliers of nectar for bees. Pollen analysis in immature honey makes possible to detect the flowers used for daily hive activities that are not usually present in the stored mature honey because the honeybees consume them during the period of expansion of the colony. On the other hand, mature honey allows to identify the pollen of the flowers that were chosen to be used as stores for the periods that there are no flowers in the environment, which is the harvest time of the beekeepers. Méndez and others(30) stated that the importance of determining the flora represented in immature honey lies in detecting the plants used early by bees and generally not represented in the harvested mature honey, as their nectar is consumed during the growth period of the hive. However, Salgado and Pire(11) and Salgado and others(31) valued these plants as a beekeeping resource.
Finally, we constructed and defined the terms “Convertible Flora” and “Support Flora”. The first term is the flora that bees will store inside the hive (Stored honey). This flora serves as store for the period when there are no blooms inside the hives and simultaneously meets three conditions: 1) Melissopalynological studies reported pollen from this flora in mature honey; 2) we reported that it remained in bloom for more than one season, and 3) we found that their flowering scale was abundant or very abundant. When worker honeybees are overexposed with flora they will accumulate only four or five flowering species in the hive(12). We defined “Support Flora” as those floral species found in immature honey in melissopalynological studies in the literature, and that during this study remained in bloom for a short period and were not abundant (less than five individuals).
2.3 Statistical analysis
We created an evaluation matrix with all the collected data. We used the Vegan: community ecology package for Shannon Index(32). To explain the factor fixed (Dependent variable) flowering, the random factors (Variables) = seasonality (autumn, spring, summer and winter), abundance, and periodicity were assessed with the Generalized Linear Models (GLM) function using maximum significance as partition criterion. This model allowed the use of non-normal distributions of errors (such as binomials, for ex. presence or absence of blooms) and non-constant variances. In this study, there was a binary response: the presence of flowers (1) and absence of flowers (0); therefore, the use of GLM is justified. All the statistical and graphical analyzes were made with program R V.3.3.2(33).
3. Results
This study indicated the diversity with Shannon Index (H) was: site 1: 3.5878, site 2: 3.6513, and site 3: 3.6689. The average value was 3.6160, and the variance 0.001051. The total number of species that bloomed showed no significant difference between each site (P= 0.9679). We observed site one = 45 species, site two = 48 species, site tree = 46 species.
We identified 48 species distributed in 28 families of Angiosperms. Those with the highest number of representatives (58% of the total) were Asteraceae (8 species), Myrtaceae (6 species), Fabaceae (3 species), Rhamnaceae (3 species), Anacardiaceae (2 species), Apiaceae (2 species), Cactaceae (2 species), Bignoniaceae (2 species); the remaining (42 %) was represented by 20 families with one representative (Table 1).
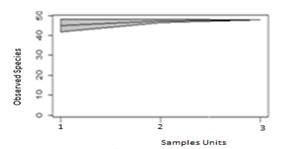
The species accumulation curve is shown in Fig.1.
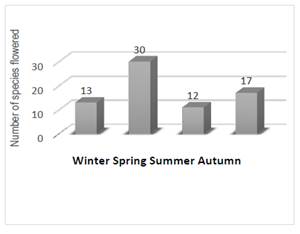
We assessed 91.7% of the taxa to native species, and the remaining 8.3% to exotic ones (Table 1, Supplementary Material). The flower calendar (Figure 2) begins in winter (27%) with only few species flowered, then spring (63%), where the number of species flowered increased abruptly, reaching the maximum. Flowering species dropped down during summer (25%), and rose again in autumn (35%), when a second flowering occurred.
We found significant differences between spring and autumn but there were not significant differences in summer (P= 0.86296) and winter (P= 0.45406).
We found in blooms (P<0.05) by species = Aloysia gratissima (P=0.172 x10-3), Baccharis trimera (P=0.376 x10-5), Baccharis articulate (P=0.633 x10-5), Blepharocalyx salicifolius (P= 0.977 x10-3), Jodina rhombifolia (P=0.0241), Eugenia uniflora (P=0.172 x10-3), Baccharis articulata (P= 7.96 x10-5), Schinus engleri (P=0.01012), Celtis tala (P=0.010), Eryngium pandanifolium (P=0.0121), and Scutia buxifolia (P=0.010).
We were able to establish the flowering time (P) (the time they remained in bloom throughout the seasons of the year) for the species identified. The species that stayed in bloom for less than one season were: Ammi visnaga, Baccharis dracunculifolia, Heterothalamus alienus, Berberis laurina, Clytostoma callistegioides, Tillandsia arequitae, Opuntia arechavaletae, Ipomoea cairica, Calliandra tweedii, Abutilon pauciflorum, Eucalyptus globulus, Myrrhinium loranthoides, Psidium cattleianum, Ligustrum lucidum, Clematis campestris, Condalia buxifolia, Fagara sp., Xylosma tweediana, and Vitex megapotamica (Table 1). On the other hand, all of the species that stayed in bloom for more than two seasons were shrubs, except Baccharis (L) (P<0.05).
We found 15 abundant species and two very abundant ones but species that significantly differed (P<0.05) were: Lithraea brasiliensis, Baccharis articulata, Baccharis trimera, Blepharocalyx salicifolius, Eugenia uniflora, Oxalis sp., Jodina rhombifolia, Aloysia gratissima, Colletia paradoxa, Scutia buxifolia.
We presented research by regional authors from subtropical environments (Argentina, Brazil and Uruguay) in Table 1Supplementary Material. We characterized the 48 species according to whether their pollen was found in immature and/or mature honeys.
We could not find for nine of the species detected in our survey any melissopalynological studies. They were: Baccharidastrum triplinervium, Berberis laurina, Clytostoma callistegioides, Tillandsia arequitae, Styrax leprosus, Cypella herbertii, Fagara sp., Vitex megapotamica and Psidium cattleianum.
| Family | Specie | Spring | Summer | Autumn | Winter | A1 | P2 | BF3 | Pr (˃ ІzІ) 4 |
| Anacardiaceae | Lithraea brasiliensis | 4 | 4 | C | * | ||||
| Schinus engleri | 3 | 3 | S | * | |||||
| Apiaceae | Ammi visnaga | 3 | 1 | SS | 0.40887 | ||||
| Eryngium pandanifolium | 3 | 3 | S | * | |||||
| Asteraceae | Baccharidastrum triplinervium | 3 | 2 | O | 0.36248 | ||||
| Baccharis articulata | 4 | 4 | C | *** | |||||
| Baccharis dracunculifolia | 4 | 1 | SS | 0.67236 | |||||
| Baccharis punctulata | 2 | 2 | S | * | |||||
| Baccharis salicifolia | 4 | 3 | SS | 0.9999 | |||||
| Baccharis trimera | 5 | 5 | C | *** | |||||
| Heterothalamus alienus | 3 | 1 | SS | 0.36248 | |||||
| Senecio brasiliensis | 3 | 2 | SS | 0.31965 | |||||
| Berberidaceae | Berberis laurina | 1 | 1 | O | 0.65684 | ||||
| Bignoniaceae | Clytostoma callistegioides | 4 | 1 | O | 0.16400 | ||||
| Dolichandra unguis-cati | 2 | 3 | MP | ** | |||||
| Boraginaceae | Echium plantagineum | 1 | 3 | SS | 0.67236 | ||||
| Bromeliaceae | Tillandsia arequitae | 3 | 1 | O | 0.67236 | ||||
| Cactaceae | Opuntia arechavaletae | 2 | 1 | SS | 0.16400 | ||||
| Wiginsia sp. | 4 | 2 | SS | 0.16400 | |||||
| Celastraceae | Maytenus ilicifolia | 1 | 3 | S | * | ||||
| Convolvulaceae | Ipomoea cairica | 3 | 1 | SS | 0.67236 | ||||
| Styracaceae | Styrax leprosus | 2 | 2 | O | . | ||||
| Euphorbiaceae | Sebastiania klotzschiana | 4 | 2 | SS | 0.67236 | ||||
| Fabaceae | Caesalpinia gilliesii | 1 | 2 | MP | . | ||||
| Calliandra tweedii | 1 | 1 | MP | 0.67236 | |||||
| Mimosa pigra | 4 | 2 | SS | 0.63927 | |||||
| Iridaceae | Cypella herbertii | 4 | 2 | O | 0.98204 | ||||
| Malvaceae | Abutilon pauciflorum | 1 | 1 | S | * | ||||
| Myrtaceae | Blepharocalyx salicifolius | 4 | 4 | C | ** | ||||
| Eucalyptus globulus | 1 | 1 | SS | 0.06803 | |||||
| Eugenia uniflora | 4 | 4 | C | ** | |||||
| Myrceugenia glaucescens | 1 | 2 | MP | 0.67236 | |||||
| Myrrhinium loranthoides | 3 | 1 | MP | 0.40887 | |||||
| Psidium cattleianum | 2 | 1 | O | 0.67236 | |||||
| Oleaceae | Ligustrum lucidum | 1 | 1 | SS | 0.36248 | ||||
| Oxalidaceae | Oxalis sp. | 5 | 2 | C | * | ||||
| Ranunculaceae | Clematis campestris | 3 | 1 | SS | 0.11522 | ||||
| Rhamnaceae | Colletia paradoxa | 4 | 2 | C | * | ||||
| Condalia buxifolia | 3 | 1 | SS | 0.99987 | |||||
| Scutia buxifolia | 4 | 2 | C | * | |||||
| Rutaceae | Fagara sp. | 2 | 1 | O | 0.22637 | ||||
| Santalaceae | Jodina rhombifolia | 4 | 3 | C | * | ||||
| Salicaceae | Xylosma tweediana | 3 | 1 | SS | 0.67236 | ||||
| Sapindaceae | Allophylus edulis | 2 | 3 | S | * | ||||
| Thymelaeaceae | Daphnopsis racemosa | 2 | 3 | S | * | ||||
| Cannabaceae | Celtis tala | 3 | 4 | S | * | ||||
| Verbenaceae | Aloysia gratissima | 4 | 3 | C | *** | ||||
| Lamiaceae | Vitex megapotamica | 3 | 1 | O | 0.67236 |
On the other hand, Schwab and others(28) and Paleologos and others(29) demonstrated a positive relationship between plant diversity and sustainability entomofauna, allowing to fulfill the ecological functions that ensure stability and resilience of insects. Santos and others(34) indicated that in Uruguay bees should not have protein problems in their diet when pollen supply is heterogeneous; that is, it comes from different flowers. Our data showed a diverse landscape with a high number of floral species, distributed in several families that spread their blooms throughout the year, with a diverse offer of nectar and pollen, which should guarantee a nutritional balance for bees.
The flowering behavior in terms of the family differed significantly (P= 7,594 x10-06). Many of the identified species belonged to the entomophilic botanical family. In this study, the most appropriate flower supply was Asteraceae, Myrtaceae, Rhamnaceae and Fabaceae. Sánchez and Lupo(35) also cited them as essential families in the flora studies of beekeeping in the Sierras de Santa Bárbara (Native Flora, Argentina). We found Asteraceae and Myrtaceae contributed throughout the seasons, standing out in autumn and winter respectively. Simultaneously, Rhamnaceae and Fabaceae stood out mainly in spring but did not in the entire period. These results agree with what Valtierra and Bonifacino(36) documented that Asteraceae is one of the three most abundant families in Uruguay with 77 species, and within it, Baccharis (L) is the most important genus. Other authors reported these species(24)(25) as typical and adapted bushes to the mountain areas in Lavalleja, Uruguay.
In Uruguay, the average period of availability of food resources for bees begins at the end of winter; reaching a flowering peak in spring that then gradually decreases in summer, steps up in autumn and drops almost to zero in winter. The primary honey harvest season is from late spring to late summer. For 15 years, silvoapiculture activities are associated with eucalyptus plantations for industrial purposes in Paysandú, Río Negro, Tacuarembó, Cerro Largo and Durazno departments, allowing the honey harvest to extend until late autumn.
We did not find studies about Psidium cattleianum, but Córdova(37) cited Psidium guayava in Mexican honey, referred to pollen in mature honey from bees in the region.
Flores and Sánchez(38) documented pollen of Dolichandra unguis-cati, Calliandra tweedii, Caesalpinia gilliesii, Myrceugenia glaucescens, Myrrhinium loranthoides in Meliponas´s honey (MP) but not in honeybee´s honey.
Table 1 shows the results after the species categorization as “Convertible Flora” (C) and “Support Flora” (S). The species that were abundant (P<0.05) or very abundant (P<0.05) and which during two or more seasons remained in bloom (P<0.05) were reported in mature honey melissopalynological studies and showed significant differences were: Lithraea brasiliensis, Baccharis articulata, Baccharis trimera, Blepharocalyx salicifolius, Eugenia uniflora, Oxalis sp., Colletia paradoxa, Scutia buxifolia, Jodina rhombifolia, Aloysia gratissima. On the other hand, we categorized eight species as “Support Flora” (S) according to the attributes of abundance and remaining flowering throughout the seasons (P<0.05), and reported in studies of immature honey: Schinus engleri, Maytenus ilicifolia, Eryngium pandanifolium, Baccharis punctulata, Abutilon pauciflorum, Daphnopsis racemosa, Allophylus edulis and Celtis tala. (Table 1 and Table S1 of Supplementary Material). We also counted the “Secondary Flora” (SS) when it met the “Support Flora” requirements, but the statistical analyses were not significant, because they were available in bloom for bee fleetingly in little time or quantities, but they are valued as a beekeeping resource, they are presented in Table S1 (SS).
4. Discussion
According to Margalef(39), the Shannon Index ranges from 1 to 5 and values below 2 indicate low diversity, from 2 to 3.5 indicate medium diversity, and above 3.5 indicate high diversity. The results of our study indicated high diversity landscapes (IS=3.6).
Figure 1 shows the Species Accumulation Curve. The slope of the species-sampling curve decreased, reaching zero and theoretically corresponded to the fact that the sampling effort to find new species reached zero. These results are in line with those of Brussa and Delfino(24) and Baston(25), that characterized an area of mountain ranges in Uruguay (Lavalleja) as one with a high diversity and repeated species in the landscape(25).
The Uruguay floral curve is similar to that observed by Basilio and Romero(40) in neighbor regions such as the Lower Delta of the Paraná River, by Andrada and Tellería(41) in southern regions of Argentina, such as the south of Caldenal, and by Tellería(42) in the Pampeana Argentina region. However, we determined that there were flower offers throughout the year with one peak of blooms in spring and second minor peak in autumn. This result is similar to those reported by Forcone and others(43) in honey Lower Valley of the Chubut River Argentina, with the forages spread and increased in start autumn.
In the Asteraceae family, Baccharis stood out as an essential resource, according to what was observed by Sánchez and Lupo(35) in the different beekeeping areas of Jujuy (Argentina). Baccharis sp. sources of nectar and pollen are species with herbaceous and shrub habits. Tejera and others(44) point them as one of the primary sources of nectar-pollen, and Corbella and others(45) described it in honey in hives along the Uruguayan coastal zone, as well as Daners and Tellería(46), who described it in commercialized (ripe) honey types from Uruguay. In this study, we detected some species with consecutive blooms that spanned during spring (B. dracunculifolia, B articulata, B. salicifolia, B. trimera), summer and autumn (B. articulata, B. salicifolia B. triplinervium, B. trimera, B punctulata) as well as winter (B. trimera).
Torreta and Basilia(47) documented self-incompatibility and the needs of biotic pollinators for (Dioecious) Scutia buxifolia, Schinus longifolia, and Jordina rhombifolia, and other native species; these species are attractive to bees. As well as other anemophilous species, such as Celtis tala, whose pollen was found in honey. It hints that bees are somehow attracted regardless the type of pollination of the species (Autogamy, allogamy in particular anemophilous pollination). Similarities between the pollen found in beehive honey and the native forest support this assumption. However, Fagúndez(48), Salgado and others(31) and Méndez and others(30) found anemophilous pollen in both mature and immature honey, and according to D’Albore(49) the presence in the honey of pollen from anemophilous species is attributed to the contamination of the nectar produced within the hive (Secondary contamination).
We present Celtis tala as a "Support Flora" (P <0.05) despite being anemophilia, since it remains blooming in spring, autumn and winter and its presence in honey has been reported as indicated above, with values ranging from 0.4 22.1%, the majority (63%) of the studies detected less than 1% of occurrence, which the bee can collect through pollen and its presence in honey could result from secondary contamination within the colony.
Asteraceae and Fabaceae were the most represented families in honey pollen within the studied area. These results agree with studies carried out by Burgos and Sánchez(50), Méndez and others(30) in Argentina for immature honey, and Sánchez and Lupo(35) in mature honey.
Salgado and others(31), Daners and Tellería(46), Corbella and others(45), Bazzuro and others(51), Sánchez and Lupo(35), as well as Fagúndez(48) mentioned Baccharis sp. pollen type in mature Apis mellifera honey in different frequency classes.
Méndez and others(30) studied the pollen in immature honey Junglas of Jujuy (Argentina) and identified as the dominant pollen the Scutia sp. and Condalia sp., which were censed in the area of study. Salgado and others(31) found in honey from the phytogeographic province of Chaco, Scutia buxifolia pollen and ranked it as a vital nectariferous resource for hives with a dominant secondary presence in mature honey.
Basilio and Romero(40) highlight the apicultural value of secondary species as an early resource for the hive, allowing the development and increase of its population.
We found 17 species reported in both mature and immature honey studies. These were Schinus engleri, Eryngium pandanifolium, Baccharis dracunculifolia, Baccharis articulata, Baccharis punctulata, Baccharis salicifolia, Baccharis trimera, Senecio brasiliensis, Mimosa Pigra, Opuntia arechavaletae, Sebastiania klotzschiana, Abutilon pauciflorum, Blepharocalyx salicifolius, Ligustrum lucidum, Colletia paradoxa, Allophylus edulis, Celtis tala.
Jato and others(52) reported the change in the use of support flora for reserve by hives that went through unfavorable weather, and after beginning the storage period, fact also indicated by Basilio and Romero(40).
Our results hinted that spring and autumn are two probable peaks of honey harvest by bees. The use of early resources that support beehive survival is called “Support Flora”. Some species that play this role were identified (P<0.05): Schinus engleri, Maytenus ilicifolia, Eryngium pandanifolium, Baccharis punctulata, Abutilon pauciflorum, Daphnopsis racemosa, Allophylus edulis and Celtis tala. These results highlight the importance of the flora survey and the melissopalynological analysis in mature and immature honey. The combination of these tools will help to correctly evaluate the environments for apiculture, according to the occurrence of species of beekeeping interest. Otherwise, potentially interesting ecosystems for apicultural activities could be disregarded.
5. Conclusions
The landscape of native forest in Lavalleja was determined to be a homogeneous zone with high diversity (Shannon Index 3.6) and high availability of food resources for bees. These zones are a proper niche for honeybee breeding and apiculture.
The vegetation survey carried out in the three study sites allowed to determine 48 species, belonging to 28 botanical families. The best represented in species diversity were Asteraceae, Myrtaceae, Rhamnaceae and Fabaceae.
There were 10 species characterized as “Convertible Flora”: Lithraea brasiliensis, Baccharis articulata, Baccharis trimera, Blepharocalyx salicifolius, Oxalis sp., Eugenia uniflora, Colletia paradoxa, Scutia buxifolia, Jodina rhombifolia, Aloysia gratissima. All of them significantly differed during spring and autumn in terms of blooming. The species classified as “Support flora” were eight: Schinus engleri, Eryngium pandanifolium, Baccharis punctulata, Maytenus ilicifolia, Abutilon pauciflorum, Allophylus edulis, Daphnopsis racemosa, Celtis tala.
From the analysis, it appears that native species constitute the leading resource throughout the beekeeping season. The beekeeping potential of the area resides in the native flora, mainly in the woody species. In addition, the flowering supply period was extensive compared to other regions of Uruguay. The strategy followed to survey the flora of beekeeping interest allows another approach: to move towards a more effective use of food resources for beekeeping. Hives are expected to be more productive when the presence of “Support and Convertible flora” is known, as proper management strategies can be developed. In such way, the suitability of different environments for developing apicultural activities can be compared, as well as the types and diversity of honey that can be harvested.
Acknowledgments
Especial thanks to Miguel Pimienta, for the hives, and Roberto Castañe, who helped in the data collection during fieldwork. This work was funded by FPTA_INIA 320 project.
References
1. Seeley TD. The wisdom of the hive: the social physiology of the honeybee colonies. Cambridge: Harvard University Press; 1995. 295p.
2. Keller L. Behavioral plasticity: levels of sociality in bees. Curr Biol. 2015;13(16):644-5.
3. Nicolson S. Bee food: the chemistry and nutritional value of nectar, pollen, and mixtures of the Two. Afr Zool. 2011;46:197-204.
4. Brodschneider R, Crailsheim K. Nutrition and health in honeybees. Apidologie. 2010;41:278-94.
5. Amaya M. Memoria y Aprendizaje en la escogenia floral de las abejas. Acta Biol Colomb. 2009;14(2):125-36.
6. Waser N. Flower constancy: definition, cause, and measurement. Am Nat.1986;127:593-603.
7. Simcock NK, Gray HE, Wright GA. Single amino acids in sucrose rewards modulate feeding and associative learning in the honeybee. J Insect Physiol. 2014;69:41-8.
8. Seeley TD. Social foraging in honeybees: how nectar foragers assess their colonys nutritional status. Behav Ecol Sociobiol. 1989;24:181-99.
9. Gruter W, Farina M. Nectar distribution and its relation to food quality in honeybee (Apis mellifera) colonies C. Insectes Soc. 2007;54:87-94.
10. Forcone A. Fuentes de néctar y polen utilizadas por Apis mellifera en el valle inferior del río Chubut (Argentina) [doctoral’s thesis]. Bahía Blanca: Universidad Nacional del Sur; 2002. 220p.
11. Salgado CR, Pire SM. Análisis polínico de las mieles del noroeste de la provincia de Corrientes (Argentina). Darwiniana. 1998;36:87-93.
12. Crane E. Honey: comprehensive survey. London: Heinemann; 1975. 645p.
13. Beekman M, Ratniek FLW. Long-range foraging by the honeybee, Apis mellifera L. Funct Ecol. 2000;14:490-6.
14. Núñez JA, Giurfa M. Motivation and regulation of honeybee foraging. Bee World. 1996;77(4):182-96.
15. Núñez JA. Honeybee foraging strategies at a food source in relation to its distance from the hive and the rate of sugar flow. J Apic Res. 1982;21(3):134-50.
16. Ghazoul J. Floral diversity and the facilitation of pollination. Ecology. 2006;94:295-304.
17. Steffan-Dewenter I, Münzenberg U, Bürger C, Thies C, Tscharntke T. Scale dependent effects of landscape context on three pollinator guilds. Ecology. 2002;83:1421-32.
18. Potts S, Vulliamy B, Dafni A, Ne'eman G, Willmer P. Linking bees and flowers: how do floral communities structure pollinator communities? Ecology. 2010;84(10):2628-42.
19. Mcintosh R. Concept and Terminology of Homogeneity and Heterogeneity. In: Kolasa J, Pickett STA, editors. Ecological Heterogeneity. New York: Springer; 1991. p. 24-46.
20. Lázaro A, Totland O. Local floral composition and the behaviour of pollinators: attraction to foraging within experimental patches. Ecol Entomol. 2010;35:652-61.
21. Neuman M, Starlinger F. The significance of different indices for stand structure and diversity in forest. Forest Ecol Manag. 2001;145:91-106.
22. Louveaux J, Maurizio A, Vorwohl G. Methods of Melissopalinology. Bee World. 1978;59:39-157.
23. Peel MC. Updated world map of Koppen Geiger climate classification. Hydrol Earth Syst Sci. 2007;11:1633-44.
24. Brussa C, Delfino L. Flora y Vegetación del Uruguay: fitogeografía y formaciones vegetales: caracterización y relaciones en el contexto biorregional. In: Memorias del VII Congreso Nacional y VI Congreso Internacional de Profesores de Biología. Tacuarembó: Asociacion de profesores de biologia; 2004. p. 65-72.
25. Baston J. Estudio de la flora arbórea de los bosques de las Sierras de las Ánimas [grade’s thesis]. Montevideo (UY): Universidad de la República, Facultad de Agronomía; 1983. 135p.
26. Chao A, Chazdon RL, Shen TJ. A new statistical approach for assessing similarity of species composition with incidence and abundance data. Ecol Lett. 2005;8:148-59.
27. Pla L. Biodiversidad: inferencia basada en el índice de Shannon y la riqueza. Interciencia. 2006;31(8):583-90.
28. Schwab A, Dubois D, Fried P, Edwards P. Estimating the biodiversity of hay meadows in north- eastern Switzerland on the basis of vegetation structure. Agric Ecosyst Environ. 2002;93:197-209.
29. Paleologos M, Bonicatto M, Marasas M, Sarandón S. Abundancia y diversidad de la coleopterofauna edáfica asociada a la cobertura vegetal y al monte cercano en viñedos tradicionales de la costa de Berisso, Buenos Aires. In: Actas del lII Congreso Brasilero de Agroecología; V Seminario Internacional sobre Agroecología; I Seminario Estatal sobre Agroecología. Porto Alegre: Embrapa; 2004. 4p.
30. Méndez M, Sánchez A, Flores F, Lupo L. Análisis polínico de mieles inmaduras en el sector oeste de las yungas de Jujuy (Argentina). Bol Soc Argent Bot. 2016;51(3):449-62.
31. Salgado CR, Piesko G, Tellería MC. Aporte de la melisopalinología de la flora melífera de un sector de la Provincia Fitogeográfica Chaqueña. Bol Soc Argent Bot. 2014;49:513-24.
32. Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O’Hara RB, Simpson GL, Solymos P, Stevens MHH, Szoecs E, Wagner H. Vegan: community ecology package [Internet]. R package version 2.5-6. 2019 [cited 2021 mar 22]. Available from: http://bit.ly/2OV9pwk.
33. R Development Core Team. R: a language and environment for statistical computing [Internet]. Vienna: R Foundation for Statistical Computing; 2016 [cited 2021 mar 22]. Available from: https://bit.ly/31v5U2G.
34. Santos E, Invernizzi C, García E, Cabrera C, Di Landro R, Saadoun A, Daners G. Contenido de proteína cruda del polen de las principales especies botánicas utilizadas por las abejas melíferas en Uruguay. Agrociencia Uruguay. 2009;13(2):9-13.
35. Sánchez A, Lupo L. Tipos polínicos presentes en mieles uniflorales de la provincia de Jujuy, Argentina. In: Memorias del 1er. Congreso Argentino de Apicultura; 2006 Jul 28 – 29; Cordoba, Argentina. Cordoba: INTA; 2006. p. 53-5.
36. Valtierra M, Bonifacino J. Revisión taxonómica de Baccharis Sect. Heterothalamus (Less.) (Asteraceae: Asterae) en Uruguay. Bol Soc Argent Bot. 2014;49(4):613-20.
37. Córdova C. Determinación geográfica y botánica de miel de abeja (Apis mellifera L.) del estado de Tabasco, México [master’s thesis]. Tabasco (MX): Institución de Enseñanza e Investigación en Ciencias Agrícolas; 2009. 147p.
38. Flores F, Sánchez A. Primeros resultados de caracterización botánica de mieles de tetragonisca angustula Latreille (Apidae, Meliponinae) criadas en la localidad Los Naranjos - Orán – Salta. Bol Soc Argent Bot. 2010;45:81-91.
39. Margalef R. Homage to Evelyn Hutchinson, or why there is an upper limit to diversity. Trans Conn Acad Arts Sci. 1972;44:211-35.
40. Basilio A, Romero E. Contenido polínico en las mieles de la región del Delta del Paraná (Argentina). Darwiniana. 1996;34(1-4):113-20.
41. Andrada AC, Tellería MC. Pollen collected by honeybees (Apis mellifera L.) from south of protein content. Grana. 2005;44:115-22.
42. Tellería MC. Plantas de importancia apícola del Distrito Oriental de la Región Pampeana (Argentina). Bol Soc Argent Bot. 1995;30:131-6.
43. Forcone A, Aloisi PV, Ruppel S, Muñoz M. Botanical composition and protein content of pollen collected by Apis mellifera L. in the north-west of Santa Cruz (Argentinean). Grana. 2011;50:30-9.
44. Tejera L, Invernizzi C, Daners G. Población y recursos alimenticios en colonias de Apis mellifera en Uruguay. Arch zootec. 2003;62:607-10.
45. Corbella E, Tejera L, Cernuschi F. Calidad y origen botánico de mieles del noreste de Uruguay. Rev INIA. 2005;(3):6-7.
46. Daners G, Tellería C. Native vs. introduced bee flora: a palynological survey of honeys from Uruguay. J Apic Res. 1998;37:221-9.
47. Torreta JP, Basilia A. Dispersión polínica y éxito reproductivo de cuatro especies arbóreas de un bosque xerófito de Argentina. Rev Biol Trop. 2009;57(1-2):283-92.
48. Fagúndez GA. Estudio de los recursos nectaríferos y poliníferos utlizados por Apis mellifera en diferentes ecosistemas del Departamento Diamante (Entre Ríos, Argentina) [doctoral’s thesis]. Bahía Blanca (AR): Universidad Nacional del Sur; 2011. 285p.
49. D'Albore GR. Methods for the characterization of the botanical and geographical origin of some bee products and for their quality control. In: Mizrahi A, Lensky Y, editors. Bee Products. Boston: Springer; 1997. p. 31-40.
50. Burgos MG, Sánchez AC. Preferencias alimenticias en las mieles inmaduras de Apis mellifera en el Chaco Serrano (Jujuy, Argentina). Bol Soc Argent Bot. 2014;49:41-50.
51. Bazurro D, Díaz R, Sánchez M. Tipificación de miel de palma butiá (Butia capitata) durante la floración de 1995-1996 en el Departamento de Rocha. Rocha: Universidad de la República; 1996. 28p. (Documentos de Trabajo; 12).
52. Jato MV, Iglesias MI, Rodríguez-Gracia V. Una contribución a la Relación ambiental de los espectros de polen de mieles de Ourense. Grana. 1994;33(4-5):260-7.
53. Costa MC, Vergara-Roig VA, Kivatinitz S. A melissopalynological study of artisanal honey produced in Catamarca (Argentina). Grana. 2013;52(3):229-37.
54. Tellería MC, Salgado C, Andrada C. Rhamnaceae asociada a mieles fétidas en Argentina. Rev Mus Argent Cienc Nat. 2006;8(2):237-41.
55. Cabrera MM. Caracterización polínica de las mieles de la provincia de Formosa, Argentina. Rev Mus Argent Cienc Nat. 2006;8:135-42.
56. Cabrera M, Andrada A, Gallez L. Floración de especies con potencial apícola en el Bosque Nativo Formoseño, Distrito Chaqueño Oriental Argentina. Bol Soc Argent Bot. 2013;48:477-91.
57. Gutiérrez B, Quiroz D. Estudio melisopalinologico de dos mieles de la porción Sur del Valle de México. Polibotánica. 2007;23:57-75.
58. Faricelli M, Kraus T, Bianco C. Análisis palinológico de las especies melitófilas de la familia Fabaceae del centro de la Argentina: Parte 1. FAVE Secc Cienc agrar. 2004;3:14-23.
Supplementary material
| Family | Specie | O | F | Bee review according to pollen present in mature honey (M), in immature honey (I) o in both (M/I), in meliponas honey(MP), no date=Sd |
| Anacardiaceae | Lithraea brasiliensis | N | 9 | (M)(46) |
| Schinus engleri | N | 8 | (M)(31)(35)(46)(48)(53)(54), (M/I)(30) | |
| Apiaceae | Ammi visnaga | E(1) | 7 | (M)(31)(46)(48)(53)(54)(55)(56) |
| Eryngium pandanifolium | N | 8 | (M)(31)(45)(46)(48)(54)(55)(56), (M/I)(44) | |
| Asteraceae | Baccharidastrum triplinervium | N | 8 | Sd |
| Baccharis articulata | N | 8 | (M)(31)(35)(40)(45)(46)(48), (I)(44), (M/I)(30) | |
| Baccharis dracunculifolia | N | 10 | (M)(31)(35)(40)(46)(48)(53), (I)(44),(M/I)(30) | |
| Baccharis punctulata | N | 4 | (M)(31)(35)(40)(45)(46)(53)(57),(I)(44),(M/I)(30) | |
| Baccharis salicifolia | N | 11 | (M)(31)(35)(40)(45)(46)(53),(I)(44),(M/I)(30) | |
| Baccharis trimera | N | 14 | (M)(31)(35)(45)(46)(53),(I)(44),(M/I)(30)(40)(44) | |
| Heterothalamus alienus | N | 8 | (M)(46),(MI)(44) | |
| Senecio brasiliensis | N | 8 | (M)(31)(38)(46)(48)(53)(54),(I)(30) | |
| Berberidaceae | Berberis laurina | N | 1 | Sd |
| Bignoniaceae | Clytostoma callistegioides | N | 9 | Sd |
| Dolichandra unguis-cati | N | 5 | (MP)(35) | |
| Boraginaceae | Echium plantagineum | N | 1 | (M)(40)(45)(48)(54) |
| Bromeliaceae | Tillandsia arequitae | N | 6 | Sd |
| Cactaceae | Wiginsia sp. | N | 10 | (I)(30) |
| Opuntia arechavaletae | N | 5 | (M)(53), (I)(30) | |
| Celastraceae | Maytenus ilicifolia | N | 1 | (M)(48)(53) |
| Convolvulaceae | Ipomoea cairica | E | 4 | (M)(57) |
| Euphorbiaceae | Sebastiania klotzschiana | N | 9 | (M)(46)(48), (M/I)(30) |
| Fabaceae | Calliandra tweedii | N | 1 | (MP)(35) |
| Mimosa pigra | N | 9 | (M)(46)(53)(55), (I)(30) | |
| Caesalpinia gilliesii | N | 1 | (M)(46), (MP)(52)(58) | |
| Salicaceae | Xylosma tweediana | N | 6 | (M)(30)(31) |
| Iridaceae | Cypella herbertii | N | 10 | Sd |
| Malvaceae | Abutilon pauciflorum | N | 1 | (M)(31)(48), (I)(35) (En trazas), (MI)(30) |
| Myrtaceae | Blepharocalyx salicifolius | N | 11 | (M)(11), (I)(30) |
| Eucalyptus globulus | E | 1 | (M)(30)(31)(38)(46)(48)(53)(55) | |
| Eugenia uniflora | N | 9 | (M)(31)(55) | |
| Myrceugenia glaucescens | N | 1 | (MP)(35) | |
| Myrrhinium loranthoides | N | 6 | (MP)(35) | |
| Psidium cattleianum | N | 3 | (M)(34) | |
| Oleaceae | Ligustrum lucidum | E | 1 | (M)(48)(53)(54), (I) (30) |
| Oxalidaceae | Oxalis sp. | N | 13 | (M)(31)(35) |
| Rhamnaceae | Scutia buxifolia | N | 9 | (M)(30)(31)(35)(45)(46)(48)(54) |
| Colletia paradoxa | N | 9 | (M)(30), (M/I)(35) | |
| Condalia buxifolia | N | 5 | (M)(30)(35)(53) | |
| Ranunculaceae | Clematis campestris | N | 7 | (M)(30)(35)(48)(53) |
| Rutaceae | Fagara sp. | N | 5 | Sd |
| Santalaceae | Jodina rhombifolia | N | 12 | (M)(48) |
| Sapindaceae | Allophylus edulis | N | 3 | (M)(31)(35)(46), (M/I)(30) |
| Styracaceae | Styrax leprosus | N | 4 | Sd |
| Thymelaeaceae | Daphnopsis racemosa | N | 5 | (M)(51) |
| Ulmaceae | Celtis tala | N | 8 | (M)(35)(46)(55), (M/I)(30) |
| Verbenaceae | Aloysia gratissima | N | 8 | (M)(31)(48)(53)(54)(55)(56) |
| Vitex megapotamica | N | 8 | Sd |
Author notes
heihoracio@gmail.com
Additional information
Author contribution
statement: RD:
conceptualization, data collection, analysis, article writing; SN, VC:
conceptualization, analysis and manuscript review. HH: conceptualization analysis and draft writing, revising and
editing.
Data transparency: Data
were available. The entire data set supporting the results of this
study was published in the article itself.
Editor: The following editor approved this article.
Valentina Mujica (https://orcid.org/0000-0002-9820-2879)
Instituto Nacional de Investigación
Agropecuaria (INIA), Montevideo, Uruguay.
Alternative link
http://agrocienciauruguay.uy/ojs/index.php/agrociencia/article/view/426/867 (pdf)

