Abstract: TRAP sequence describes the chronic perfusion of an “acardiac” twin by a “pump” twin through permeable intertwined vascular channels. It occurs in 1 in 35,000 births or 1 in 100 monozygotic twin pairs. It is diagnosed by ultrasound findings of a normally developing fetus and an amorphous mass often with discernible fetal parts. Colour Doppler reveals reverse blood flow to the acardiac twin within the umbilical artery leading to typical complications. Expectant management is reasonable in the absence of poor prognostic features. The purpose of this article is to review the basics and current status of this condition, with emphasis on diagnosis and expectant management.
Keywords: twin reversed arterial perfusion sequence, acardiac twin, monochorionic twins.
Resumen: TRAP describe la perfusión crónica de un gemelo “acardíaco” por un gemelo “de bomba” a través de canales vasculares entrelazados permeables. La secuencia TRAP ocurre en 1 de cada 35.000 nacimientos o en 1 de cada 100 pares de gemelos monocigóticos. Se diagnostica mediante los hallazgos ecográficos de un feto de desarrollo normal y una masa amorfa con frecuencia con partes fetales perceptibles. El Doppler color revela el flujo sanguíneo reverso hacia el gemelo acardíaco dentro de la arteria umbilical lo que lleva a las complicaciones típicas del cuadro. El manejo expectante es razonable en ausencia de características pronósticas deficientes. El propósito de este artículo es revisar los aspectos básicos y el estado actual de esta condición, haciendo énfasis en el diagnóstico y el manejo expectante.
Palabras clave: secuencia de perfusión arterial reversa en gemelos, gemelo acárdico, gemelos monocoriónicos.
Casos Clinicos
Twin Reverse Arterial Perfusion Sequence (TRAP). Case report on expectant management.
Received: 06 October 2020
Accepted: 10 December 2020

The twin reversed arterial perfusion (TRAP) sequence consists in a combination of a normal appearing twin, called the pump twin, that pumps blood to an abnormal co-twin who usually does not have a functional heart, namely the acardiac twin1. The TRAP sequence is an anomaly unique to monochorionic gestations2,3.
It is estimated to have an incidence of 1% in monochorionic twin pregnancies and 1 per 35,000 births2,4. Its importance lies in the poor prognosis of the surviving twin despite its low incidence4. En el estudio de la placenta es habitual encontrar las inserciones de los cordones umbilicales muy cercanas entre sí y siempre se observa la comunicación arterio-arterial, además de una veno-venosa, ambas superficiales4. Up to one third of fetuses have an abnormal karyotype2.
The aetiology of the TRAP sequence is not precisely known. Altered organogenesis in response to reverse flow through abnormal arterio-arterial or veno-venous anastomoses has been proposed, leading to premature hypoxia and impaired cardiac development2,10, resulting in deoxygenated blood from the umbilical artery of the ‘’pump twin’’ flowing retrograde into the umbilical arteries (or artery because there is often only one) and iliac vessels of the acardiac twin, thus perfusing the lower body with deoxygenated blood to a much greater extent than the upper body, resulting in a highly anomalous twin mass2,3. The result is a spectrum of malformations, shrinkage anomalies of previously existing tissues and incomplete morphogenesis of tissues mainly in the upper body3,6. Acardiac twins are true parasites that are totally dependent on the pump twin for their blood supply and have no functional placental tissue of their own5. The pump twin that perfuses itself and also the acardiac twin develops congestive heart failure with fetal hydrops and polyhydrohydramnios, which usually ends in premature delivery or death4.
The acardiac twin can endanger the well-being of the pump twin by three different mechanisms: First, the acardiac twin mass as it grows increases intrauterine volume and, consequently, the risk of preterm delivery; the mean gestational age at delivery is 31 weeks. Second, the phenomenon of vascular steal can lead to high output heart failure and hydrops with polyhydramnios, and even intrauterine death. Finally, the acardiac twin further deoxygenates the already deoxygenated blood of the pump twin, this deoxygenated blood returns through a veno-venous anastomosis to the pump twin and further reduces its oxygen level, causing chronic hypoxia and growth restriction5,10.
Prenatal diagnosis is based on the recognition of one fetus with a normal appearance and another fetus with a very abnormal appearance or an amorphous mass of tissue in a monochorionic pregnancy2. Color Doppler shows blood flow in the acardiac mass and its umbilical cord with reversed direction of blood flow towards the acardiac twin2,6,10. The acardiac twin is incompatible with life regardless of its extent, whereas the prognosis in the anatomically healthy pump twin varies considerably10.
The aim of this article is to review the basic aspects and current status of this condition, focusing mainly on diagnosis and expectant management.
Case report
The following case presented is of a 35-year-old female patient from Cochabamba, a school teacher, with a history of a previous cesarean section six years earlier, with no other medical history of importance, whose condition began during her second pregnancy, after an ultrasound examination in week 13 which reported: apparent biamniotic monochorionic twin pregnancy with one fetus normal and the other possibly acardiac. In view of this finding, two other studies were performed by two other doctors who reported a twin pregnancy with one fetus alive and the other one deceased.
The morphological study performed at 19 weeks, in private practice, already concluded as: biamniotic monochorionic twin pregnancy with a normal fetus and the other one acardiac. With this result the patient went to the Gynecology and Obstetrics Service of the Hospital Obrero No. 2, from where she was referred to the Fetal Medicine Unit, which she attended only in the 21st week. At the unit, a new ultrasound study was performed and the following findings were found: TRAP sequence in biamniotic monochorial twin gestation with a normal living fetus (pump fetus) without hemodynamic compromise and the other fetus was acardiac (Figure 1).
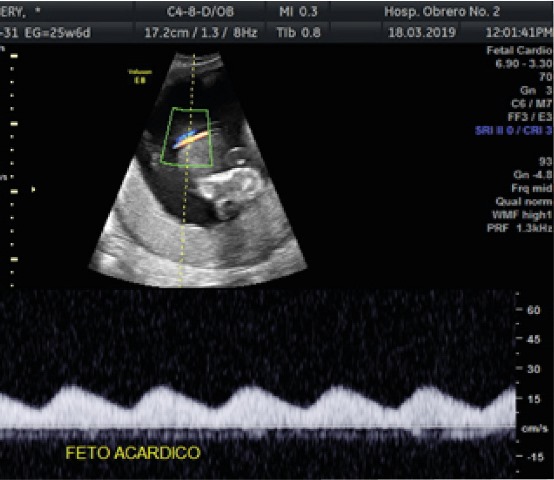
Since her gestational age was far from the possibility of fetal survival and there was no hemodynamic impairment at that time, she was asked to undergo strict periodic controls only after the 24th week, looking for signs of cardiac deterioration in the pump fetus and volume control of the acardiac fetus.
As instructed, she went for consultation in the 24th week of pregnancy; the study performed showed a healthy fetus with 798 grams with no signs of pump failure on Doppler study; the volume of the acardiac fetus was 1612 cc. Due to the acardiac fetal volume, as a sign of poor prognosis, the patient was asked to go to a referral center outside the country for occlusion of the umbilical cord of the acardiac fetus. When the patient refused the proposed trip, she was offered the alternative of expectant management with strict surveillance of the living fetus looking for signs of heart failure and measurement of the growth of the acardiac fetus, with the possibility of termination of the pregnancy if markers of pump failure were found.
By week 25 the ultrasound study showed the acardiac fetus with a volume of 1660 cc and the healthy fetus with 990 grams with mild dilatation of the free umbilical vein and intrahepatic, suprahepatic veins, which were interpreted as initial venous stagnation. Tricuspid flow and ductus venosus were normal, as was the amniotic fluid. Fetal lung maturation and Doppler controls were started twice a week. Consultation with cardiology was requested for an eventual digitalization if venous stagnation persisted.
By week 27, a slight dilatation of the right atrium, suprahepatic veins and umbilical vein was observed. Maternal digitalization was started with 0.25 mg/day after an attack dose. Atrial dilatation improved, but the umbilical vein remained mildly dilated.
By week 28, improvement of the sonographic findings of venous stasis was noted. By week 29 the fetal weight of the pump twin was 1466 grams and the volume of the acardiac fetus was 2,200 grams; Doppler parameters remained stable.
By week 30 the patient started with uterine contractions, cervical modifications and neck shortening; the estimated fetal weight at that time was 1817 grams, with no signs of pump failure, normal amniotic fluid; the acardiac fetus had a volume of approximately 2569cc. An emergency cesarean section was scheduled with the diagnosis of 30.5 week pregnancy, TRAP sequence in monochorionic biamniotic twin pregnancy with one live fetus and one acardiac fetus, onset of labor. The cesarean section resulted in a live normal female fetus weighing 1700 grams and an amorphous acardiac twin weighing 2300 grams, both within the weight range calculated by ultrasound. (Figure 2).
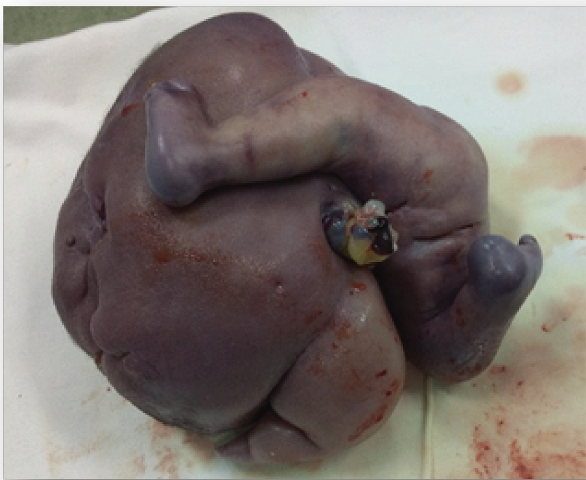
The newborn was admitted to neonatal therapy for one week and admitted to the neonatal unit for 54 days, after which he was discharged.
The patient was discharged on the third day. No autopsy of the acardiac twin was performed due to the refusal of the mother. Pathological anatomy of the placenta was reported as monochorionic biamniotic with abnormal vascular connections.
Of the many malformations of acardial twins, the most frequent type presents a well-developed, though often deformed, pelvis and lower limbs and an absent or rudimentary upper body.
In rare cases it appears simply as an amorphous mass5; in our case, only the lower limbs were recognizable, the rest was a shapeless mass.
Predicting the prognosis of the donor twin depends on the ratio of the weight of the acardiac twin to the weight of the healthy twin, with a 30% chance of congestive heart failure when the ratio is greater than 0.7, compared to 10% when the ratio is less than 0.755.
Given that the size of the acardiac twin is an important prognostic factor, this parameter was measured in all ultrasound examinations; likewise, the other criteria for poor perinatal outcome such as polyhydramnios, Doppler indices, hydrops in the donor twin, cardiomegaly, pericardial effusion, tricuspid insufficiency, reverse flow in the ductus venosus, pulsatility in the umbilical vein2,5,7, were closely monitored. The weight of the acardiac twin, in our case, was always higher than 0.7 with respect to the normal fetus, corroborated after cesarean section.
Expectant management, described as reasonable in the absence of poor prognostic features, has had good survival rates when the weight ratio between the acardiac twin and the pump twin is 50% or less. If expectant management is performed, weekly surveillance, including fetal echocardiography, is recommended, looking for signs of fetal decompensation in the pump twin7.
In our case, expectant management was proposed in view of the impossibility of performing umbilical cord occlusion due to technical resources and in the absence of signs of cardiac decompensation in the normal twin.
Medical management with maternal administration of digoxin or indomethacin has been reported. The use of maternal digitalis to treat heart failure in the pump twin was reported by Simpson et al. in 1983; no subsequent reports of this therapy with digoxin have been reported. Ash et al. (1990) reported the use of indomethacin in an acardiac pregnancy complicated by polyhydramnios at 21 weeks due to the high risk of preterm delivery8. The case of twin pregnancy with TRAP sequence in our service was managed with maternal digitalization at full dose due to dilatation of the umbilical vein, suprahepatic and right atrium, which was taken as initial cardiac pump failure, with apparent good response at 72 hours.
In case of poor prognostic characteristics far from term, it is recommended to consider the interruption of vascular communication between the twins, between 16 and 26 weeks in hospital institutions with material and technical resources. The indications for in utero treatment are the following: hemodynamic compromise of the pump twin, either by finding fetal hydrops or by alteration of echocardiographic and fetal Doppler parameters consistent with cardiac insufficiency and size ratio between both fetuses, reporting that if it is greater than 70% premature delivery occurs in 90% of the cases and cardiac insufficiency in 30%; however, estimating the weight of the acardiac twin is not very accurate. It has been postulated that it would be more advantageous to measure the relationship between the abdominal circumference of both fetuses, which is more reproducible4. In our case, the fetal volume and the estimated fetal weight were close to the real one.
Current treatment modalities include umbilical cord occlusion of the acardiac twin by bipolar cord coagulation, laser ablation and radiofrequency ablation, which is not available in our institution. The optimal therapeutic approach has not yet been definitively determined7. The technique consists in occluding the cord of the acardiac fetus with laser or bipolar in the free cord or at the base of implantation with radiofrequency or monopolar coagulation; it is advisable to perform it at 16-18 weeks before hydrops appears. Reported survival rates are between 80 and 90%9.
In general, the natural history of the disease is death by cardiac failure of the pump twin in 35-55% of cases, therefore, when a diagnosis of TRAP sequence is made, the patient should be recommended to go to a referral center that has experience in “inutero” treatment.
In environments such as ours, since we do not have this type of centers, we can offer the alternative of expectant management with strict control of fetal cardiac function, control of amniotic fluid, volume of the acardiac fetus and fetal weight of the normal twin. If there is suspicion of pump failure in the normal twin in pregnancy far from fetal viability, as defined by the Neonatology Service (in our service it is 26 weeks), maternal digitalization may be indicated under the supervision of a cardiologist; amniodrainage in case of severe polyhydramnios, until the fetus reaches a gestational age with reasonable expectation of viability.



