Cardiac arrhythmia following spinal anaesthesia at caesarean section
Arritmia cardiaca posterior a anestesia espinal en cesárea
Gaceta Médica Boliviana, vol. 43, no. 1, 2020
Universidad Mayor de San Simón
Casos clinicos
Received: 07 October 2019
Accepted: 20 March 2020

Abstract: Arrhythmia and tachycardia are very common in pregnant women, although spinal block may be a safe anaesthetic technique, severe tachycardia, cardiac arrest and other arrhythmias are reported during spinal anaesthesia practices. Bupivacaine and levobupivacaine may increase the PR interval and QRS duration and prolong cardiac conduction. Dexmedetomidine has sympatholytic, sedative, respiratory stability without ventilatory depression, amnesic and analgesic properties, adverse effects of dexmedetomidine are initial hypertension, hypotension, nausea, bradycardia, atrial fibrillation, pulmonary edema, oliguria and thirst. The patient is 33 years old and 39.2 weeks pregnant, with no history of pathology. She received spinal anaesthesia and after a 10 min bradycardia of 39 corrected with atropine and later with sinus arrhythmia. In conclusion, the presence of cardiac arrhythmias may be due to several factors, and continuous monitoring of the electrocardiogram is essential to recognise and correct them in a timely manner.
Keywords: sinus arrhythmia, dexmedetomidine, spinal anesthesia.
Resumen: La arritmia y taquicardia son muy comunes en las embarazadas, aunque el bloqueo espinal pueda ser una técnica anestésica segura, la taquicardia grave, la parada cardiaca y otras arritmias son relatadas durante las prácticas de raquianestesia. La bupivacaina y levobupivacaina pueden aumentar el intervalo PR y la duración del QRS y prolongar la conducción cardiaca. La dexmedetomidina tiene propiedades simpaticolíticas, sedativas, estabilidad respiratoria sin depresión ventilatoria, amnésicas y analgésicas, los efectos adversos de la dexmedetomidina son la hipertensión inicial, hipotensión, naúseas, bradicardia, fibrilación atrial, edema pulmonar, oliguria y sed. Paciente de 33 años de edad con embarazo de 39,2 sem. Sin antecedentes patológicos. Recibe anestesia espinal y a los 10 min bradicardica de 39 corregida con atropina y posterior con arritmia sinusal. En conclusión la presencia de arritmias cardiacas puede deberse a varios factores, es trascendental la vigilancia y monitoreo continuo del electrocardiograma para reconocer y corregir de manera oportuna.
Palabras clave: arritmia sinusal, dexmedetomidina, anestesia espinal.
Cardiac arrhythmias are a major cause of morbidity and mortality during the perioperative period, as well as in critically ill patients in intensive care units (ICU)1.
Los agentes anestésicos pueden presentar actividad pro-arritmica y antiarritmica al inducir actividad eléctrica por medio de varios mecanismos2.
Anaesthetic agents can exhibit both proarrhythmic and anti-arrhythmic activity by inducing electrical activity through various mechanisms2.
Neuroaxial anaesthesia is considered the preferred method of obstetric analgesia and anaesthesia for vaginal or caesarean delivery care. The effects of drugs to the foetus are avoided, anaesthesiologists generally do not use sedatives and other drugs before birth3.
Arrhythmia and tachycardia are very common in pregnant women, although spinal block may be a safe anaesthetic technique, severe tachycardia, cardiac arrest and other arrhythmias are reported during spinal anaesthesia practices4.
Bupivacaine and levobupivacaine may increase the PR interval and QRS duration thereby prolonging cardiac conduction5.
Dexmedetomidine is a potent and highly selective high-selective α-2-adrenoceptor agonist with sympatholytic, sedative, respiratory stability without ventilatory depression, amnesic and analgesic properties, which has already been described as a useful and safe supplement in several clinical applications6 .
Adverse effects of dexmedetomidine include initial hypertension, hypotension, nausea, bradycardia, atrial fibrillation, pulmonary oedema, oliguria and thirst7. Overdose can cause first- or second-degree atrioventricular block. Most of the adverse effects associated with the use of dexmedetomidine occur during or immediately after the attack dose8 .
Case presentation
Paciente A 33 year old female patient from Cercado - Cochabamba, on 03 June 2019 goes to the Gynecology-Obstetrics emergency department of the Hospital Obrero N° 2 “Caja Nacional de Salud”.
Patient is 39, 2 weeks pregnant weeks gestation due to the last menstrual period (FUM), previous Caesarean section, obstetric formula: G2C1P0A0. With mild abdominal pain.
The Obstetrics and Gynaecology Department decided to terminate the pregnancy with the diagnosis of pregnancy at 39.2 weeks, previous caesarean section and prodromal labour, and requested an assessment by anaesthesiology. The patient reported that she was fasting for more than two hours and the pre-anaesthetic evaluation is detailed below:
Personal background
No relevant pathological history and no allergies.
Personal habits: no alcoholism, smoking.
Surgical history: bilateral tonsillectomy 22 years ago under general anaesthesia without complications, caesarean section 6 years ago for circular cord with spinal anaesthesia without complications.
Gynaeco-obstetric history: G2 C1 P0 A0.
Drugs: ferrous sulphate 200 mg, 1 tablet orally daily.
General Physical Examination:
Patient with anxiety, afebrile, with moist and pink mucous membranes, hemodynamically stable.
Vital signs: blood pressure 110/70 mmhg. Heart rate 80 beats per minute. Oxygen saturation at room temperature 96%.
Regional physical examination:
Airway: Incomplete teeth, Mallampati I, Thyro-mental distance greater than 6 cm. Sternal-mental distance greater than 10 cm.
Neck: Mobile, symmetrical.
Neurological: conscious, oriented. Glasgow: 15/15.
Heart: rhythmic, regular.
Lungs: preserved vesicular murmur.
Abdomen: globose at the expense of a pregnant uterus, with a uterine height of 29 cm. Single product, in longitudinal position, cephalic presentation, left dorsum, fetal heart rate of 150 beats per minute and active fetal movements.
Extremities: normotrophic, normotonic with palpable peripheral pulses, capillary refill less than 2 seconds.
Supplementary examination
Hemogram and blood chemistry (Table 1)

Obstetric ultrasound: placenta anterior, not previa. Grade I. (Figure 1)
It does not have an initial electrocardiogram as per service protocol.
Diagnostic Impression:
9.2 weeks pregnant
Previous caesarean section
Prodromes of labour
Single live fetus
American Society of Anesthesiologists Physical Status Classification ASA-II
Anesthetic plan: neuraxial anaesthesia, post-surgical analgesia, prevention of hypotension, nausea and vomiting.
The patient in the operating room underwent basic monitoring of vital signs, and was found to have BP: 112/62 mmhg, HR: 76x’, SatO2: 96%. Patient in sedentary position, asepsis and antisepsis were performed, lumbar puncture by median approach with Whitacre pencil point spinal needle No. 27 at the level of the L4-L5 space; confirming the correct placement of the needle by the free flow of cerebrospinal fluid, anaesthetic mixture was administered containing: 10 mg of 0.5% hyperbaric Bupivacaine and dexmedetomidine 3 µg. Patient in supine position with pelvic wedge for uterine displacement to the left. Basic monitoring of non-invasive blood pressure, continuous electrocardiogram, heart rate and pulse oximetry was performed; these parameters were checked every 3 minutes trans-surgically.
Dermatome level reached T5. Ten minutes after anaesthesia, mean arterial pressure increased by 6 mmHg from baseline and bradycardia of 39 beats per minute, atropine 0.5 mg IV was administered, and BP 100/58 mmhg. With a good result of 105 beats per minute and maintained throughout the anaesthetic procedure between 108 - 95 beats per minute (Figure 2).
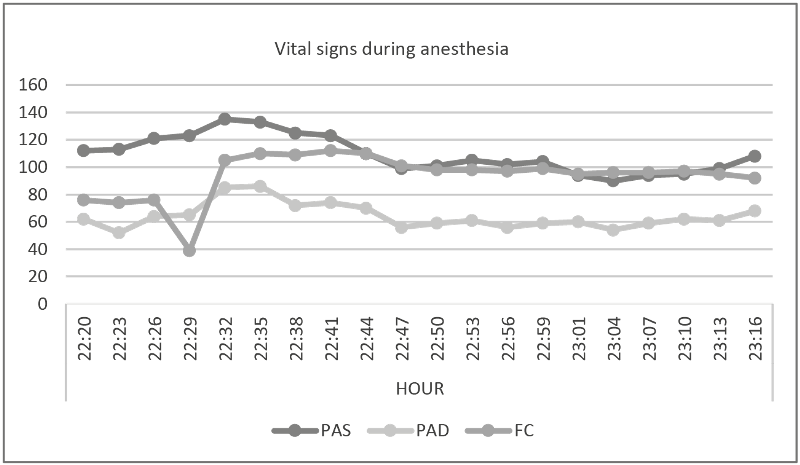
Crystalloid solutions were administered at a rate of 15ml/kg, a female newborn was obtained with an APGAR of 8 at one minute and APGAR 9 at 5 minutes, and oxytocin 3 IU was administered intravenously as a bolus in three minutes.
Our patient presented an increase in blood pressure within the first 10 minutes.
Alterations in the electrocardiogram were evidenced without repercussions on blood pressure (Figure 3). At the conclusion of the procedure, a cardiological assessment was requested
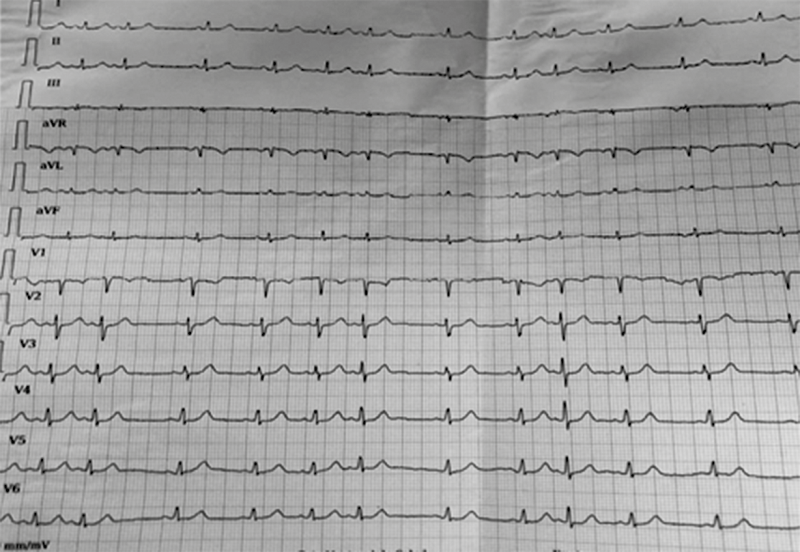
The cardiology service mentioned that it was a transient sinus arrhythmia that only warranted monitoring, without medication. The patient was discharged two days after surgery without any cardiac problems, with sinus rhythm and was monitored by obstetrics in 10 days.
Discussion
The presentation of the clinical case is important to determine the causes of bradycardia, especially in obstetric anaesthesia.
Regional anaesthesia is the preferred technique for caesarean section because it allows maternal-fetal contact without airway manipulation and maintenance of respiratory reflexes. According to Joseph et al9, recognised adverse effects of regional anaesthesia in parturients and non-parturients are hypotension, arrhythmia and cardiac arrest.
Cardiac arrhythmias are a major cause of morbidity and mortality during the perioperative period, as well as in critically ill patients in intensive care units. Rhythm disturbances can be well tolerated by the normal heart. However, it can cause significant haemodynamic instability in patients with a congenital or acquired heart problem1.
In the clinical case presented, the patient had no cardiac abnormalities on monitoring and visualisation of the electrocardiogram at the start of anaesthesia. It is important to remember that hormonal and haemodynamic changes that occur during pregnancy may also result in pro-arrhythmic effects. Pregnancy may trigger the development of new arrhythmias or aggravate existing ones. In addition, levobupivacaine and bupivacaine for spinal anaesthesia were shown to shorten QT intervals in patients. Premature atrial and ventricular beats are also very frequent4.
We can mention that the patient presents with anxiety and as described by Deniz et al4, Sato et al10 and Jang11, triggering factors for bradycardia are: stress, emotions, pain, anxiety, fear, blood phobia, coughing, urination and increased afferent activity of the trigeminal and carotid sinus.
Shen et al12 describe surgical manoeuvres such as externalisation of the uterus can also increase vagal tone and cause arrhythmia.
Anaesthetic agents can trigger pro-arrhythmic and anti-arrhythmic activity by inducing electrical activity through various mechanisms. Bupivacaine is the most commonly used drug in obstetric anaesthesia4. In our patient bupivacaine heavy 10 mg was used. There are reports, as mentioned by Leone et al5, that the dissociation caused by bupivacaine is 10 times longer than lidocaine and the blockade induced by bupivacaine can accumulate, resulting in a more marked cardiac depression.
On the other hand, the patient had a rise in blood pressure within the first 10 minutes plus atropine administration that was elevated for a further 20 minutes, after which a drop in blood pressure was observed that ranged from 100/60 mmhg to 103/62 mmhg. Whereas, Ingersoll et al13 indicate that dexmedetomidine has a biphasic response with a transient increase in blood pressure followed by a sustained decrease in heart rate which appears to be a similar combination of a baroreflex-mediated reduction in heart rate coinciding with transient increases in blood pressure, reduced centrally mediated sympathetic tone and increased vagal tone. Talke et al14 argue that perioperative use of α-2 agonists associated with bradycardia, sedation and dry mouth and one patient in their study had sinus pause for 5-10 seconds during intubation.
The aim of using dexmedetomidine to prolong analgesic rescue and decrease the need for intravenous analgesics can be used intravenously, intranasally, intrathecally, Xiong et al15 describe dexmedetomidine which can prolong the duration of sensory blockade and motor blockade.
The newborn recorded APGAR of 8 and 9 at 1 minute and 5 minutes, Wang et al16 state that there are no adverse effects in newborns and that the placental transfer rate of dexmedetomidine is 0.68.
The dermatome level reached in the patient is T5, according to Owzuck et al17, the high sympathetic blockade may contribute to haemodynamic instability occurring with regional anaesthesia and the inotropic agents used may cause proarrhythmic effects.
At 10 minutes after intrathecal administration of the drugs, bradycardia of 39 beats per minute was present and atropine 0.5 mg was administered and as mentioned by Afonso et al8;, the common side effects of dexmedetomidine are bradycardia and hypotension, the temporary effects can be controlled with atropine, ephedrine and volume infusion.
Kalra et al18 reported bradycardia followed by atypical Wenckebach block following spinal anaesthesia in an obstetric patient.
In conclusion, the presence of cardiac arrhythmias may be due to several factors, and continuous monitoring of the electrocardiogram is essential to recognise and correct in a timely manner in patients under regional anaesthesia, perform left uterine lateralisation, prevent maternal hypotension, and administer cocarga fluids.
References
1. Choudhury, M. Dexmedetomidine: The Anesthetic as an Antiarrythmic; Cardiol Pharmacol [Internert].2015 [Citado el 27 de junio de 2019];4(3). Disponible en: https://doi.org/10.4172/2329-6607.1000153
2. Hanci V, Ayoglu H, Yurtlu S, Yildirim N, Okyay RD, Erdogan G, Sayin E, Turan IO. An evaluation of P wave dispersion, QT, corrected QT and corrected QT dispersion intervals on the electrocardiograms of malnourished adults; Anaesth Intensive Care [Internet].2010 [Citado el 27 de junio de 2019];38(1), 122–127. Disponible en: https://journals.sagepub.com/doi/pdf/10.1177/0310057X1003800120
3. Wang C, Liu S, Han C, Yu M, Hu Y, Liu C. Effect and placental transfer of dexmedetomidine during caesarean section under epidural anaesthesia; J Int Med Res [Internet]. 2017 [citado el 18 de mayo de 2019];45(3):964–972. Disponible en: https://doi:10.1177/0300060517698330
4. Deniz Y, Dilek Okyay, Volkan Hanci, Serhan Yurtlu, Hilal Ayoglu, Isil Özkoçak Turan, Efecto de la Levobupivacaína y la Bupivacaína en las Dispersiones de la Onda P, QT y QT Corregido (QTc) en Cesárea; Rev Bras Anesthesiol [Internet].2013 [Citado el 26 de junio de 2019];63(2):202. Disponible en: https://doi.org/10.1016/j.bjanes.2012.04.005
5. Leone S, Di Cianni S, Casati A, Fanelli G.Pharmacology, toxicology, and clinical use of new long acting local anesthetics, ropivacaine and levobupivacaine; Acta Biomed [Internet].2008 [Citado el 02 de julio de 2019];79(2), 92–105. Disponible en: http://www.embase.com/search/results?subaction=viewrecord&from=export&id=L352771924%0Ahttp://www.actabiomedica.it/data/2008/2_2008/leone.pdf
6. Carollo D, Nossaman B, Ramadhyani U. Dexmedetomidine: A review of clinical applications; Current Opinion in Anaesthesiology [Internet].2008 [Citado el 28 de junio de 2019]; 21(4), 457–461. Disponible en: https://doi.org/10.1097/ACO.0b013e328305e3ef
7. García Botero Andrés, Rodríguez Leonardo, Salazar Pérez Félix Arturo, Venegas Saavedra Alberto. Use of Dexmedetomidine in Total Intravenous Anesthesia (TIVA); Rev Colomb Anestesiol [Internet]. 2011 [Citado el 27 de junio de 2019]; 39(4): 514-526. Disponible en:http://www.scielo.org.co/scielo.php?script=sciarttext&pid=S0120-33472011000400005&lng=en. http://dx.doi.org/10.5554/rca.v39i4.70.
8. Afonso Joana, Reis Flávio. Dexmedetomidina: rol actual en anestesia y cuidados intensivos; Rev. Bras. Anestesiol. [Internet]. 2012 [Citado el 18 de junio de 2019]; 62(1):125-133. Disponible en: http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0034-70942012000100015&lng=en. https://doi.org/10.1590/S0034-70942012000100015
9. Joseph SE, Minehart RD. Third-degree heart block during spinal anesthesia for cesarean delivery; AA Case Rep [Internet].2014 [Citado el 27 de junio de 2019];3(1):3-5. Disponible en: https://doi.org/10.1213/XAA.0000000000000028
10. Sato S, Nakamori E, Kusumoto G, Shigematsu K, Yamaura K. Neurally Mediated Syncope During Cesarean Delivery: A Case Report; AA Case Rep [Internet]. 2017 [Citado el 02 de julio de 2019];8(5):96-99. Disponible en: https://doi.org/10.1213/XAA.0000000000000440
11. Jang YE, Do SH, Song IA.Vasovagal cardiac arrest during spinal anesthesia for Cesarean section; Korean J Anesthesiol [Internet].2013 [Citado el 27 de junio de 2019];64(1), 77–81. Disponible en: https://dx.doi.org/10.4097/kjae.2014.64.1.77
12. Shen CL, Ho YY, Hung YC, Chen PL. Arrhythmias during spinal anesthesia for Cesarean section; Can J Anaesth [Internet].2000 [Citado el 27 de junio de 2019];47(5):393-397. Disponible en: https://doi:10.1007/BF03018966
13. Ingersoll-Weng Esperanza, Manecke Gerard, Thistlethwaite Patricia. Dexmedetomidine and Cardiac Arrest; Anesthesiology [Internet].2004 [Citado el 20 de febrero de 2020];100(3), 738–39. Disponible en: https://doi.org/10.1097/00000542-200403000-00037
14. Talke Pekka, Chen Richard, Thomas Brian, Aggarwall Anil, Gottlieb Alexandru, Thorborg Per, Heard Stephen, Cheung Albert, Son Stanley, Kallio Antero. The hemodynamic and adrenergic effects of perioperative dexmedetomidine infusion after vascular surgery; Anesthesia and Analgesia [Internet].2000 [Citado el 23 de febrero de 2020]; 90(4), 834-39. Disponible en: https://doi.org/10.1097/00000539-200004000-00011
15. Xiong Yaqin, Ma Yushan, Wu Lan, Luo Dong. Case Report Intravenous dexmedetomidine as an adjuvant in anesthetic management of a paturient for cesarean section with uncontrolled hyperthyroidism; Int J Clin Exp Med [Internet].2016 [Citado el 27 de junio de 2019];9(8),16913–17. Disponible en: https://pdfs.semanticscholar.org/b39e/6baea5e61554ee92e410489d53d53613b28d.pdf
16. Wang Changsheng, Liu Shijiang, Han Chuanbao, Yu Min, Hu Youli, Liu Cunming. Effect and placental transfer of dexmedetomidine during caesarean section under epidural anaesthesia; J Int Med Res [Internet].2017 [Citado el 18 de mayo de 2019];45(3):964-972. Disponible en: https://doi.org/10.1177/0300060517698330
17. Owczuk Radoslaw, Sawicka Wioletta, Wujtewicz, Magdalena Anna, Kawecka Aleksandra, Lasek Jerzy, Wujtewicz Maria. Influence of spinal anesthesia on corrected QT interval; Regional Anesthesia and Pain Medicine [Internet].2005 [Citado el 03 de junio de 2019];30(6),548–52. Disponible en: https://doi.org/10.1016/j.rapm.2005.06.005
18. Kalra Seema, Hayaran Nitin. Case Report Arrhythmias following spinal anesthesia for cesarean delivery - Is Wenckebach common? J Anaesthesiol Clin Pharmacol [Internet].2011[Citado27 de junio de 2019];27(4),541–44. Disponible en: https://doi.org/10.4103/0970-9185.86604
Author notes
E-mail: wonderful122.paa@gmail.com
Alternative link
http://www.scielo.org.bo/scielo.php?script=sci_arttext&pid=S1012-29662020000100015&lng=es&nrm=iso (html)

