INTRODUCTION
The largest number of protected areas (PA) in the world is in Brazil (1), totaling more than 30000 Km2 (2). About 43% of Brazilian Amazon are considered PA (3). Although there are doubts about the purpose of conservation units, studies indicate that these are useful in reducing deforestation and ichthyofauna conservation (4,5).
The Amazon basin is home to the world's largest ichthyofauna diversity (6,7). In the Madeira River basin, more than 820 species of fish were recently identified (8). In the Machado River basin, Madeira River sub-basin, 48 species were collected at the Pirarara and Tamarupa rivers (9), 140 species in small streams (10) and 74 species in the Tarumã river, middle Machado River (11). The studies on evaluation of fish stocks in the Amazon region are scarce (12), as well as information on fish morphometric relationships (13,14).
The length-weight relationship (LWR) has many functions in the area of fish ecology (15). The LWR allows i) to predict fish length based on weight and vice versa, ii) calculate the growth rate of the species and iii) obtaining the body state of the individuals sampled (16). The LWR studies are necessary to understand the fish life cycle, especially in regions where fishing represents the important economic activities and the main animal protein consumed by traditional communities (12). The objective of this study was to calculate the LWR of 13 species of fish captured in the Tarumã River, Jaru Biological Reserve, as the first study on the biology of fish species in this PA.
MATERIALS AND METHODS
The Jaru Biological Reserve (Rebio Jaru) is administered by the Instituto Chico Mendes de Conservação da Biodiversidade (ICMBio). The Rebio Jaru hydrographic network (9°27’19” S, 61°40’43” W - 9°47’04” S, 61°40’19” W) is part of the Machado River basin, Rondônia State, Brazil. The Tarumã River is located in the Rebio Jaru area.
We performed collections of freshwater fish in May and September 2015. We collected the fish using a total of eight gillnets with meshes of 2 x 20 m (mesh sizes: 30, 40, 50, 60, 70, 80, 90 and 100 mm, opposite knots), these were euthanized in a solution of clove oil (17). The fish were fixed in 10% formalin solution, preserved in 70% ethanol and later deposited in the Coleção de Peixes da UFMT, Brazil. The fish sampling was authorized by the ICMBio (license code: 48723–2/2015).
The individuals captured were identified with the help of identification keys (8) and biometric data (total length (TL) in cm and total weight (TW) in g) were recorded. Weight and length were estimated using the expression P = aCb, where P = weight total, C = total length, a = intercept and b = angular coefficient (18); the parameters a and b were estimated after logarithmic transformation of weight and length data (19). We verified in the fishBase (20) the absence of the LWR records, as well as the occurrence of the TL and TW maximum values recorded for the species captured in the Tarumã river. Statistical tests were performed using the R-package (21) with significance of p<0.05.
RESULTS
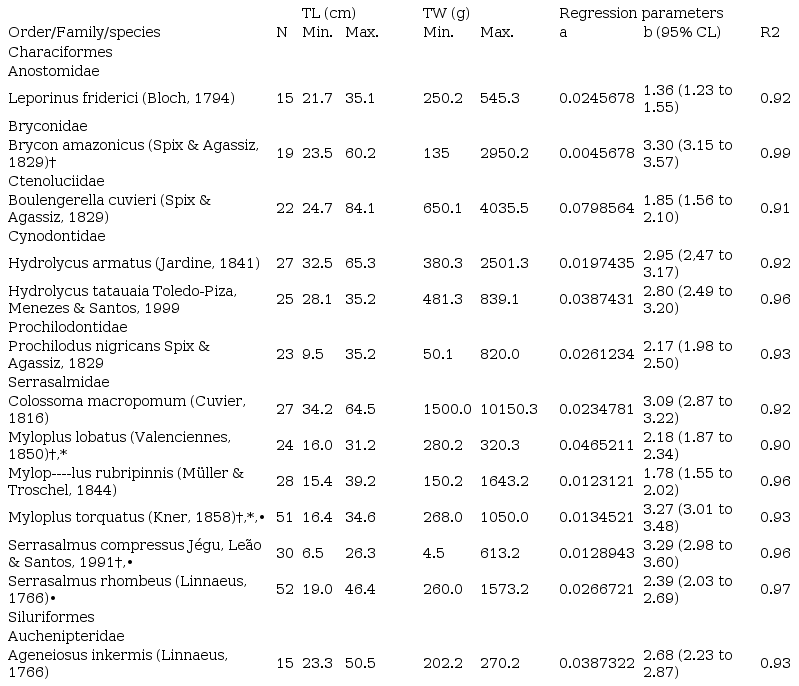
The LWR was estimated from 343 specimens belonging to thirteen species,
seven families and two orders (Table 1). The linear regressions were
significant (p≤0.001). The R2 varied from 0.91 in Boulengerella cuvieri to 0.99 in Brycon amazonicus. The intercept a varied from 0.0045678 in B. amazonicus to 0.0798564 in B. cuvieri (mean 0.0422121±0.0532370). The value of b varied from 1.36 in Leporinus friderici to 3.30 in B. amazonicus (mean 2.33±1.37).
The specie Colossoma macropomum had an isometric growth
pattern (b=3.09) (Table 1).
Table 1
Descriptive statistics and
length–weight relationship parameters for 13 fish species, Tarumã
River, Jaru Biological Reserve, Amazon. New length-weight relationships (*), maximum total weight (†) and maximum total length (•). Systematic positions were based on Nelson et al. (2016).
| |
TL (cm)
| |
TW (g)
| |
Regression parameters
|
|
Order/Family/species
|
N
|
Min.
|
Max.
| |
Min.
|
Max.
| |
a
|
b (95% CL)
|
R2
|
|
Characiformes
| | | | | | | | | | |
|
Anostomidae
| | | | | | | | | | |
|
Leporinus friderici (Bloch, 1794)
|
15
|
21.7
|
35.1
| |
250.2
|
545.3
| |
0.0245678
|
1.36 (1.23 to 1.55)
|
0.92
|
|
Bryconidae
| | | | | | | | | | |
|
Brycon amazonicus (Spix & Agassiz, 1829)†
|
19
|
23.5
|
60.2
| |
135
|
2950.2
| |
0.0045678
|
3.30 (3.15 to 3.57)
|
0.99
|
|
Ctenoluciidae
| | | | | | | | | | |
|
Boulengerella cuvieri (Spix & Agassiz, 1829)
|
22
|
24.7
|
84.1
| |
650.1
|
4035.5
| |
0.0798564
|
1.85 (1.56 to 2.10)
|
0.91
|
|
Cynodontidae
| | | | | | | | | | |
|
Hydrolycus armatus (Jardine, 1841)
|
27
|
32.5
|
65.3
| |
380.3
|
2501.3
| |
0.0197435
|
2.95 (2,47 to 3.17)
|
0.92
|
|
Hydrolycus tatauaia Toledo-Piza,
Menezes & Santos, 1999
|
25
|
28.1
|
35.2
| |
481.3
|
839.1
| |
0.0387431
|
2.80 (2.49 to 3.20)
|
0.96
|
|
Prochilodontidae
| | | | | | | | | | |
|
Prochilodus nigricans Spix & Agassiz, 1829
|
23
|
9.5
|
35.2
| |
50.1
|
820.0
| |
0.0261234
|
2.17 (1.98 to 2.50)
|
0.93
|
|
Serrasalmidae
| | | | | | | | | | |
|
Colossoma macropomum (Cuvier, 1816)
|
27
|
34.2
|
64.5
| |
1500.0
|
10150.3
| |
0.0234781
|
3.09 (2.87 to 3.22)
|
0.92
|
|
Myloplus lobatus (Valenciennes, 1850)†,*
|
24
|
16.0
|
31.2
| |
280.2
|
320.3
| |
0.0465211
|
2.18 (1.87 to 2.34)
|
0.90
|
|
Myloplus rubripinnis (Müller & Troschel, 1844)
|
28
|
15.4
|
39.2
| |
150.2
|
1643.2
| |
0.0123121
|
1.78 (1.55 to 2.02)
|
0.96
|
|
Myloplus torquatus (Kner, 1858)†,*,•
|
51
|
16.4
|
34.6
| |
268.0
|
1050.0
| |
0.0134521
|
3.27 (3.01 to 3.48)
|
0.93
|
|
Serrasalmus compressus Jégu, Leão & Santos, 1991†,•
|
30
|
6.5
|
26.3
| |
4.5
|
613.2
| |
0.0128943
|
3.29 (2.98 to 3.60)
|
0.96
|
|
Serrasalmus rhombeus (Linnaeus, 1766)•
|
52
|
19.0
|
46.4
| |
260.0
|
1573.2
| |
0.0266721
|
2.39 (2.03 to 2.69)
|
0.97
|
|
Siluriformes
| | | | | | | | | | |
|
Auchenipteridae
| | | | | | | | | | |
|
Ageneiosus inkermis (Linnaeus, 1766)
|
15
|
23.3
|
50.5
| |
202.2
|
270.2
| |
0.0387322
|
2.68 (2.23 to 2.87)
|
0.93
|
DISCUSSION
The new LWRs for 2 species, new maximum weight for 4 species and maximum sizes recorded for 3 species highlight the scarcity of information on biological aspects of Amazon fishes. The allometric coefficients of the L. friderici, B. cuvieri, Prochilodus nigricans, Myloplus lobatos, Myloplus rubripinnis and Serrasalmus rhombeus species are not among the values (2.7–3.5) described by Froese (16). Factors such as habitat, area, seasonality, degree of stomach repletion, sex ratio, health, sample size, reproductive stages and preservation methods, could justify the values not in line with that proposed by Froese (16,22,23,24). These results can provide information for the FishBase as well as important data for the management of icthyofauna and conservation of the Tarumã River Basin.
Conflict of interest
There is no conflict of interest
Acknowledgements
We are grateful to the staff members of the ICMBio for their financial support and technical assistance in the fieldwork and Universidade Federal de Rondônia for their laboratory support.
Referências
1. Bernard E, Penna LAO, Araújo E. Downgrading, downsizing, degazettement, and reclassification of protected areas in Brazil. Conservation Biology. 2014; 28(1):939-950. DOI: https://doi.org/10.1111/cobi.12298
2. Peres CA. Why we need megareserves in Amazonia. Conservation Biology. 2005; 19(1):728-733. DOI: https://doi.org/10.1111/j.1523-1739.2005.00691.x
3. Veríssimo A, Rolla A, Souto Maior APC, Monteiro A, Brito B, Souza C, Augusto CC, Cardoso D, Conrado D, Araújo E. Áreas protegidas na Amazônia Brasileira: Avanços e Desafios. 1ª edição. São Paulo: Imazon/ISA; 2011. URL: https://imazon.org.br/areas-protegidas-na-amazonia-brasileira-avancos-e-desafios-2/
4. Bruner AG, Gullison RE, Rice RE, Da Fonseca GA. Effectiveness of parks in protecting tropical biodiversity. Science. 2001; 291(5501): 125-128. DOI: https://doi.org/10.1126/science.291.5501.125
5. Frederico RG, Zuanon J, De Marco Jr P. Amazon protected areas and its ability to protect stream-dwelling fish fauna. Biological Conservation. 2018; 219(1):12-19. https://doi.org/10.1016/j.biocon.2017.12.032
6. Reis RE. Conserving the freshwater fishes of South America. International Zoo Yearbook. 2013; 47(1):65-70. DOI: https://doi.org/10.1111/izy.12000
7. Reis RE, Albert JS, Di Dario F, Mincarone MM, Petry P, Rocha LA. Fish biodiversity and conservation in South America. Journal of Fish Biology. 2016; 89(1):12-47. DOI: https://doi.org/10.1111/jfb.13016
8. Queiroz LJ, Torrente-Vilara G, Ohara WM, Pires THS, Zuanon J, Doria CRC. Peixes do Rio Madeira. 1ª edição. São Paulo: Dialeto Latin American Documentary; 2013.
9. Perin L, Shibatta OA, Bernarde PS. Fish, Machado River basin, Cacoal urban area, state of Rondônia, Brazil. Check list. 2007; 3(1):94-97. http://dx.doi.org/10.15560/3.2.94
10. Casatti L, Pérez-Mayorga MA, Carvalho FR, Brejão GL, Costa ID. The stream fish fauna from the rio Machado basin, Rondônia State, Brazil. Check list. 2013; 9(1):1496-1504. http://dx.doi.org/10.15560/9.6.1496
11.Costa ID, Ohara WM, Almeida M. Fishes from the Jaru Biological Reserve, Machado River drainage, Madeira River basin, Rondônia State, northern Brazil. Biota Neotropica. 2017; 17(1):1-9 . DOI: https://doi.org/10.1590/1676-0611-BN-2016-0315
12. Freitas TMS, Prudente BS, Fontoura NF, Montag LFA. Length-weight relationships of dominant fish species from Caxiuana National Forest, Eastern Amazon, Brazil. Journal of Applied Ichthyology. 2014; 30:1081-1083. DOI: https://doi.org/10.1111/jai.12436
13. Giarrizzo T, Oliveira RRS, Andrade MC, Goncalves AP, Barbosa TAP, Martins AR. Length–weight and length–length relationships for 135 fish species from the Xingu River (Amazon Basin, Brazil). Journal of Applied Ichthyology. 2015; 31(1):415-424. https://doi.org/10.1111/jai.12677
14. Schmid K, Andrade MC, Jesus AJS, Araujo JS, Santos PRB, Giarrizzo T. Length-Weight Relationships for Fish Fauna from Waterbodies in the Upper Tapajos River Basin of Palito Mountain Ridge, Brazilian Amazon Region. Biota Amazônica. 2015; 59(1):112-114. DOI: https://doi.org/10.18561/2179-5746/biotaamazonia.v5n3p112-114
15. Beyer JE. On length–weight relationships computing the mean weight of the fish of a given length class. Fishbyte.1987; 59(1):11-13.
16. Froese R. Cube law, condition factor and weight-length relationships: history, meta-analysis and recommendations. Journal of Applied Ichthyology. 2006; 22(1):241-253. https://doi.org/10.1111/j.1439-0426.2006.00805.x
17. American veterinary medical association. Report of the AVMA panel on euthanasia. Journal of the American Veterinary Medical Association. 2001; 218(1):669–696. URL: https://avmajournals.avma.org/toc/javma/218/5
18. Le Cren ED. The length - weight relationship and seasonal cycle in gonad weight and condition in the perch (Perca fluviatilis). Journal Animal Ecology. 1951; 20(1):201-219.
19. Vanzolini PE. Métodos estatísticos elementares em sistemática zoológica. 1ª edição. São Paulo: Ed. Hucitec; 1993.
20. Froese R, Pauly D. FishBase (version Feb 2018). In: Roskov Y. Ower G, Orrell T, Nicolson D, et al, editors. Species 2000 & ITIS Catalogue of Life. 2019 Annual Checklist (Digital resource at www.catalogueoflife.org/annual-checklist/2019. Vol.1. 1a ed. Leiden: Naturalis; 2019.
21. R Development core team. R: a language and environment for statistical computing, Version 3.5.2. [Internet]. 2019. URL Disponible en: http://www.r-project.org
22. Miranda R, Galicia D, Monks S, Pulido-Flores G. Weight–length relationships of some native freshwater fishes of Hidalgo State, Mexico. Journal of Applied Ichthyology. 2009; 25(1):620-621. https://doi.org/10.1111/j.1439-0426.2009.01319.x
23. Hossain MY, Rahman MM, Fulanda B, Jewel MAS, Ahamed F, Ohtomi J. Length–weight and length–length relationships of five threatened fish species from the Jamuna (Brahmaputra River tributary) River, northern Bangladesh. Journal of Applied Ichthyology. 2012; 28(1): 275-277. https://doi.org/10.1111/j.1439-0426.2011.01900.x
24. Hossain MY, Sayed SRM, Mosaddequr Rahman M, Ali MM, Hossen, MA, Elgorban AM, Ohtomi J. Length–weight relationships of nine fish species from the Tetulia River, southern Bangladesh. Journal of Applied Ichthyology. 2015; 31:967–969. https://doi.org/10.1111/jai.12823





