



Fisiología de cultivos
Mercury pollution in hornbeam (Carpinus betulus) trees in green space at industrial park area (Iran
Contaminación por mercurio en árboles de carpes (Carpinus betulus) en espacios verdes en el área del parque industrial (Irán)
Temas Agrarios
Universidad de Córdoba, Colombia
ISSN: 0122-7610
ISSN-e: 2389-9182
Periodicity: Semestral
vol. 25, no. 1, 2020
Received: 10 October 2019
Accepted: 27 December 2019

Abstract: The heavy metal mercury is a toxic metals that can accumulate in plant organs in large quantities and is toxic to humans. Hornbeam, Carpinus betulus, is a species from Caspian hyrcanian mixed forests. In this research, it was studied how much mercury hornbeam absorbed from the environment. Twenty five trees were randomly selected, and the distances between trees were 10 to 15 m in industrial park North of Iran. From each tree selected, 15 g leaves and 15 g root were harvested using horticultural scissors and 15 g of soil from 30 cm depth. Four stations were selected near steel factory. A control station located 2 km from the steel factory was selected and three stations were located near the steel factory. Samples of leaves, soil and roots are prepared after coding, placed in paper envelopes and transferred to the lab. The amount of mercury in plant samples was prepared by dry digestion with atomic absorption spectroscopy. To compare the effect of mercury concentration on leaf and root, Tukey's test was used. The average concentration of mercury in soil was 0.96 mg/kg, in root 0.551 mg/kg and leaf 0.28 mg/kg. It was observed that as mercury increases, the leaf chlorophyll content decreases. Results allowed concluding that Carpinus betulus tree accumulation amount is suitable for absorbing the contamination of mercury in soil pollution.
Keywords: Carpinus betulus, Mercury, Pollution, Root, Soil pollution.
Resumen: El mercurio es un metal tóxico que puede acumularse en los órganos de las plantas en grandes cantidades ocasionando toxicidad a los humanos. Carpinus betulus es una especies de los bosques mixtos hircanos del Caspio. En esta investigación, se estudió la cantidad de mercurio que pueden absorber los árboles de carpes del ambiente. Veinticinco árboles fueron seleccionados al azar, las distancias entre árboles fueron de 10 a 15 m en un parque industrial en el norte de Irán. De cada árbol seleccionado, se cosecharon 15 g de hojas y 15 g de raíz con tijeras hortícolas, y 15 g de tierra a partir de 30 cm de profundidad. Se seleccionaron cuatro estaciones cerca de la fábrica de acero. Se seleccionó una estación de control ubicada a 2 km de la fábrica de acero y se ubicaron tres estaciones cerca de la fábrica de acero. Las muestras de hojas, tierra y raíces se prepararon y codificaron, se colocan en sobres de papel y se transfirieron al laboratorio. La cantidad de mercurio en las muestras de plantas se preparó mediante digestión en seco con espectroscopía de absorción atómica. Para comparar el efecto de la concentración de mercurio en la hoja y la raíz, se utilizó la prueba de Tukey. La concentración promedio de mercurio en el suelo fue de 0.96 mg/kg, en la raíz 0.551 mg/kg y en la hoja 0.28 mg/kg. Se observó que con el aumento de mercurio la cantidad de clorofila en la hoja disminuye. Se puede concluir que la cantidad de acumulación de metales mercurio en el árbol Carpinus betulus es adecuada para absorber la contaminación del mercurio de la contaminación del suelo.
Palabras clave: Carpinus betulus, Contaminación, Contaminacion del suelo, Mercurio, Raiz.
INTRODUCTION
The heavy metal cadmium and lead-mercury can accumulate in plant organs in large quantities toxic to humans and animals, and at the same time, no symptoms of toxicity are present in the plant (Terry, 1999).These metals are absorbed through the roots and leaves and transmitted to animals or humans, causing metabolic disorders and not pursuing any beneficial biological purpose. Heavy metals are hazardous environmental and hazardous pollutant for living organisms, which, through entry into the food chain, create health hazards for humans, plants and living organisms (Raskin, 2000).In contaminated soils, plants that have the ability to absorb metals are known as one of the purification methods for these soils. Heavy metal deactivates many important enzymes that can disrupt photosynthesis and respiration and other metabolic processes, easily absorbed through the roots of the plant and with concentrations that are dangerous to the food chain (Yoon et al.,2006).
With concentrations that are hazardous to the food chain, it can be toxic in plant tissues at the cellular level, which can reduce growth. Therefore, preventing the absorption of these metals by the roots of plants can be a strategy to minimize the biologic effects of these elements (Bada ,2010). Generally, all plants are able to absorb heavy metals, but trees have a more effective role in absorbing metals in urban environments and can protect residential areas and human accumulation centers against their adverse effects (Terry, 1999).
Therefore, the reduction of this type of pollution through the development and expansion of vegetation is one of the methods that can contribute to the reduction of pollutants and their harmful effects (Baker,1990). Mercury disrupts the respiratory mechanism if it is accumulated in human tissues. Types of mercury cationic compounds are absorbed by soil particles; they are deposited in phosphate, carbonate, and mercury sulfur, which cannot be transported to soil profiles (Lombi et al.,2008). Today, due to the application of mercury in dental industries dental diseases, the use of mercury in tooth filling and its complications are also considered. Mercury is a toxic metal to which human beings are exposed, and even more toxic than arsenic and lead. Research shows that 80% of mercury vapor is injected from the lungs into the blood and brain and other organs and tissues of the body (Thangavel, 2004).
Mercury also causes nervous system impairment organs and cardiovascular impacts (Thangavel, 2004). Phytoremediation is an environmental and scientific technology that is suitable for developing countries; unfortunately, despite this potential, it is still not used in some countries like Iran. The green plant engineering reduces the risks of environmental pollutants such as heavy metals, rare elements, organic compounds and radioactive substances. The information available about the potential for heavy metals and other pollutants to be absorbed by plants is negligible (Sinicropi et al., 2010). Therefore, it is essential to carry out research in this field in order to introduce new methods of refining and selecting the appropriate plant species(Glick, 2003).
In studies, it has been pointed out that the high biomass and the proper adaptation conditions of plant species can play an important role in the refining of heavy metals contaminated soils (Nkansah, 2016).
The growth rate of root of a plant is one of the important indexes of plant resistance to different concentrations of a metal. Since roots are particularly susceptible to the presence of toxic metals and the first limb that is exposed to toxicity, root length is used as one of the most important criteria for the effects of metals on plant.
Mercury reduces the activity of photosynthesis, water absorption and antioxidant enzymes in the plant (Atobatele, 2015). Hornbeam (Carpinus betulus) is a species from Caspian Hyrcanian Mixed Forests. In this research, it was studied how much mercury from the environment hornbeam trees could absorb.
MATERIALS AND METHODS
Four stations were identified for the study; a control station located 2 km from the steel factory was selected and three stations were located near the steel factory. In each site, a transect was selected and samples were randomly taken from leaves, and roots. From each station, 25 trees were randomly selected; distances between selected trees were 10 to 15 m in industrial park area in North of Iran. From each tree selected, 15 g leaf tissue and 15 g root tissue were harvested using horticultural scissors, and 15 g of soil taken from 30 cm depth. Accordingly, in each locality, samples of leaves, soil and roots are prepared, after encoding, placed in paper envelopes and transferred to the laboratory. Leaf samples were carefully removed from the petiole, and a number of surface roots were harvested from the surrounding area. Leaves and surface roots of the trees were washed with water after harvest, and then dried in an oven at 70 °C. The amount of mercury after plant sample digestion was obtained by dry digestion with atomic absorption. Data obtained from plant experiments were organized in SPSS (Statistical Package for the Social Sciences) software. Data analysis was performed to determine the amount of metal accumulation in the shoot and root of the plant by analysis of variance. To compare the effect of mercury concentration on leaf and stem and root, Tukey's test was used.
Chlorophyll measurements were performed using Arnon method (Arnon, 1949). To this end, 0.05 g of leaf tissue with 5 ml acetone was mixed at a concentration of 80%. The resulting mixture was centrifuged for 10 min at 13,000 rpm. The extract was separated and measured its volume to 8 ml of chlorophyll using spectrophotometer at a wavelength of 645 nm and 663 nm, respectively.
RESULTS AND DISCUSSION
The average accumulation of mercury in the factory soil was 0.93 mg kg-1 and outside the factory was 0.6 mg kg-1. Average mercury accumulation inside the plant was higher (Figure 1). The average accumulation of mercury in the root of trees in near the steel factory was 0.551 mg kg-1 and control station was 0.28 mg kg-1. It is observed that the average concentration of mercury in soil, root, leaf in stations near steel factory, was 0.96 mg kg-1, 0.551 mg kg-1, 0.28 mg kg-1, respectively. It seems that the average mercury accumulation of soil is higher than leaf and root (Figure 1).
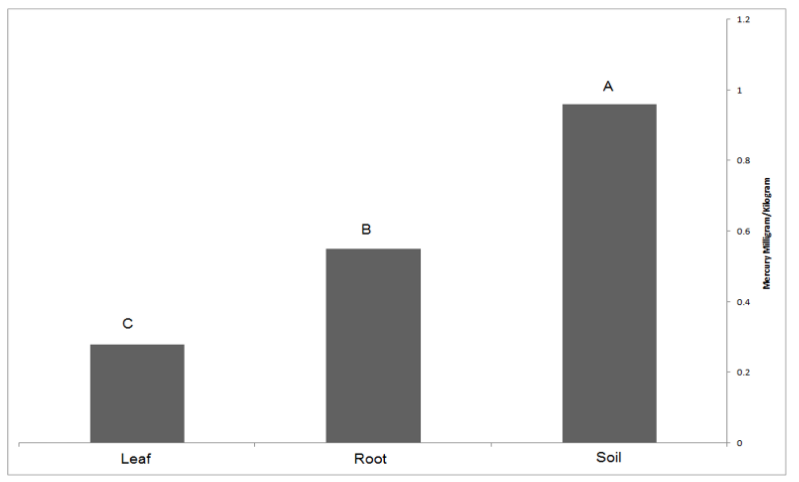
There is a significant difference between the mean mercury accumulation among soil, leaf and root of the trees inside the factory (Significant level 0.05).
It was observed that as mercury increases the amount of leaf chlorophyll content is decreases. Tukey test results show that there was a significant difference in leaf chlorophyll content inside the factory and outside the factory (Control station) (Significant level 0.05) (Figure 2).
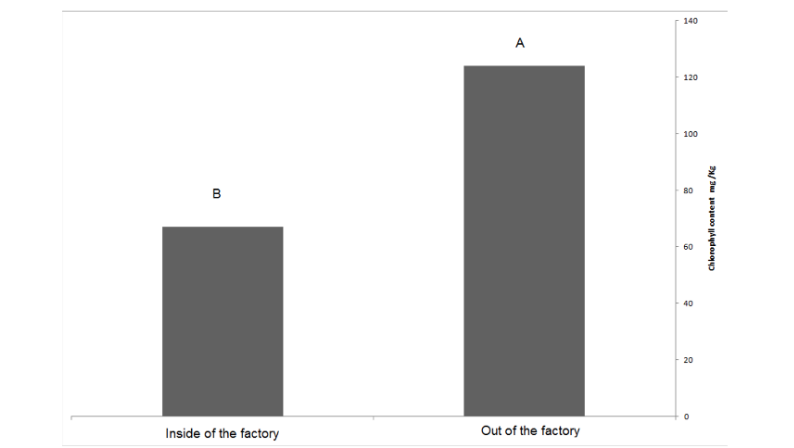
In soil, root and leaf samples, there was a significant difference between the mean of mercury accumulation near steel factory and control station of the steel factory, at significance level 0.05. Root of a plant is one of the important indexes of plant resistance to different metal concentrations. Because root is especially susceptible to the presence of toxic metals and the first limb that is exposed to toxicity, root length is used as one of the most important criteria for the effects of metals on plants. Atobatele (2015) showed that high biomass and proper adaptation conditions of plant species can play an important role in the treatment of heavy metal contaminated soils (Pant, 2015). The results indicated that mercury concentration in root tissues is higher than in leaf tisues, which is consistent with other results of studies in which the concentration of mercury in stems is less than that of the root (Raskin, 2000). Studies have shown that the efficiency of this method is appropriate for the application of plants that have high growth and high biomass. A significant reduction in the total chlorophyll content of mercury chloride treated leaves reflects the damage caused on chlorophyll biosynthesis, in which the hornbeam trees in the plant areas also showed a significant decrease in chlorophyll content in the leaf of the trees (Terry,1999).
CONCLUSIONS
According to the study, it can be concluded that based on the amount of accumulation of heavy metal mercury by Carpinus betulus trees, it is suitable for absorbing the contamination of mercury in soil pullution.
REFERENCES
Terry N., Banuelos G.S.1999. Phytoremediation of contaminated soil and water, CRC Press,p33-44.
Raskin I., Ensley B.D.2000. Phytoremediation of toxic metals., John Wiley and Sons, P.77-79.
Yoon J., Cao X., Zhou Q., Ma L.Q. 2006.Accumulation of Pb, Cu, and Zn in native plants growing on a contaminated Florida site, Sci. Total Environ. 368 : 456–464.
Bada B.S., Raji K.A.2010. Phytoremediation potential of kenaf (Hibiscus cannabinus L.) grown in different soil textures and cadmium concentrations, African J. Environ. Sci. Technol. 4:35-45
Baker A.J.M., Proctor J.1990. The influence of cadmium, copper, lead, and zinc on the distribution and evolution of metallophytes in the British Isles, Plant Syst. Evol. 173 : 91–108.
Lombi E., Zhao F.J., Dunham S.J., McGrath S.P.2001. Phytoremediation of heavy metal–contaminated soils, J. Environ. Qual. 30 : 1919–1926.
Thangavel P., Subbhuraam,C. V .2004.Phytoextraction: role of hyperaccumulators in metal contaminated soils, PROCEEDINGS-INDIAN Natl. Sci. Acad. PART B. 70: 109–130.
Sinicropi M.S., Caruso A., Capasso A., Palladino C., Panno A., Saturnino C.2010.Heavy metals: toxicity and carcinogenicity, Pharmacologyonline. 2 : 329–333.
Glick B.R., 2003.Phytoremediation: synergistic use of plants and bacteria to clean up the environment, Biotechnol. Adv. 21 : 383–393.
Nkansah M.A., Korankye M., Darko,G. Dodd M.2016. Heavy metal content and potential health risk of geophagic white clay from the Kumasi Metropolis in Ghana, Toxicol. Reports. 3 : 644–651.
Arnon D.I.1949. Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris, Plant Physiol. 24 1-16.
Atobatele O.E., Olutona G.O.2015. Distribution of three non-essential trace metals (Cadmium, Mercury and Lead) in the organs of fish from Aiba Reservoir, Iwo, Nigeria, Toxicol. Reports. 2 : 896–903.
Pant D., Sharma V., Singh P.2015. Pb detoxification in Equisetum diffusum, Toxicol. Reports. 2 : 716–720.

